Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another.
Question:
Fig. 15.3 outlines the classic scheme for separating a mixture of insoluble chloride salts from one another. Explain the chemistry involved in the various steps of the figure.
Fig. 15.3
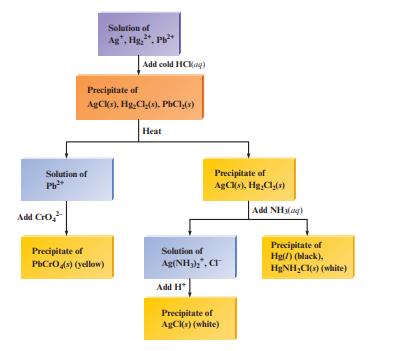
Transcribed Image Text:
Solution of Pb²+ Add CrO₂²- Solution of Ag, Hg., Pb²+ Precipitate of AgCl(s), Hg,Cl(s), PbCl(s) Precipitate of PbCrO4(s) (yellow) Add cold HCKag) Heat Precipitate of AgCl(s), Hg₂Cl(s) Solution of Ag(NH3)2, CT Add H Precipitate of AgCl(s) (white) Add NH3(g) Precipitate of Hg(/) (black). H₂NH₂Cl(s) (white)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The figure you sent me appears to show a separation scheme for a mixture of three ca...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Fig. 15.2 summarizes the classic method for separating a mixture of common cations by selective precipitation. Explain the chemistry involved with each of the four steps in the diagram. Fig. 15.2...
-
Outline a procedure for separating a mixture of benzoic acid, 4-methylphenol, aniline,and benzene using acids, bases, and organic solvents.
-
Devise a procedure for separating a mixture of the four stereoisomers of isoleucine into its four components: (+)-isoleucine, (-)-isoleucine, (+)-alloisoleucine, and (-2)-alloisoleucine (Problem 31)....
-
Each of the systems in Problems 11 through 18 has a single critical point (x 0 , y 0 ). Apply Theorem 2 to classify this critical point as to type and stability. Verify your conclusion by using a...
-
If the claimants had refuted the governments assertion that the cash was illicit, would the result in this case have been different? Why or why not?
-
The Carlberg Company has two manufacturing departments, assembly and painting. The assembly department started 10,000 units during November. The following production activity unit and cost...
-
For Skole Company, a mixed cost is \($40,000\) plus \($8\) per direct labor hour. Diagram the behavior of the cost using increments of 1,000 hours up to 5,000 hours on the horizontal axis and...
-
A town council is considering building a new bridge over a small river that runs through the town to reduce congestion on the existing bridge and reduce com-muting times. Each of 1,000 commuters who...
-
Q3-Syarikat Abu and Syarikat Hassan are 2 different rival company in Klang are offering a CNC press machine for sale to Syarikat Baba. Syarikat Abu charges RM A to deliver and install the machine....
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
Calculate the molar solubility of Cd(OH) 2 , K sp = 5.9 10 -11 .
-
Under what circumstances can you compare the relative solubilities of two salts directly by comparing the values of their solubility products? When can relative solubilities not be compared based on...
-
Would you agree or disagree with the statement that Conflict resolution through collaboration needs trust; people must rely on one another.
-
Warnerwoods Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for March. Date March 1 Activities Beginning inventory Purchase March 5 March 9...
-
Mary stands 200 meters from a water tower. She holds up a ruler 0.5 meters in front of her and measures the tower to be 0.05 meters tall. How tall is the water tower? Joe holds a two-meter stick...
-
Discuss the three main challenges you will need to consider and explain how they should be applied in design thinking and considering a viable International Business Idea.
-
Current Designs is always working to identify ways to increase efficiency while becoming more environmentally conscious. During recent brainstorming session, one employee suggested to Diane Buswell,...
-
1. Volcanic Eruption A 4,200 m tall volcano is erupting, and hot lava is spewing up into the air! Unfortunately, there is a city of people 1,600 m from the near edge of the volcano, as shown in the...
-
Do tall students tend to have better vocabulary skills than short students? We might think so looking at a sample of students from grades 1, 6, and 12 of Lake Wobegon school district. The correlation...
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
Draw a mechanism for the following transformation: NaOH, heat
-
The reaction in the previous problem is an equilibrium process. Draw a mechanism of the reverse process. That is, draw a mechanism showing conversion of the conjugated, cyclic enone into the acyclic...
-
When 2,6-heptanedione is heated in the presence of aqueous sodium hydroxide, a condensation product with a six-membered ring is obtained. Draw the product and show a mechanism for its formation.
-
During this module, we covered a variety of relevant cost concepts that managers including cost accountants and managerial accountants face in the real world. These topics come up on almost a daily...
-
From the listing provided below, select (v) for those which are considered to be red flags that fraud may have occurred and (X) for others. ?? 1. no internal auditing staff 2. weak internal controls...
-
how to do the entry in sage 50 for Transfer $5 000 from Bank: Credit Card to Bank:Chequing Account

Study smarter with the SolutionInn App


