How is acid strength related to the value of Ka? What is the difference between strong acids
Question:
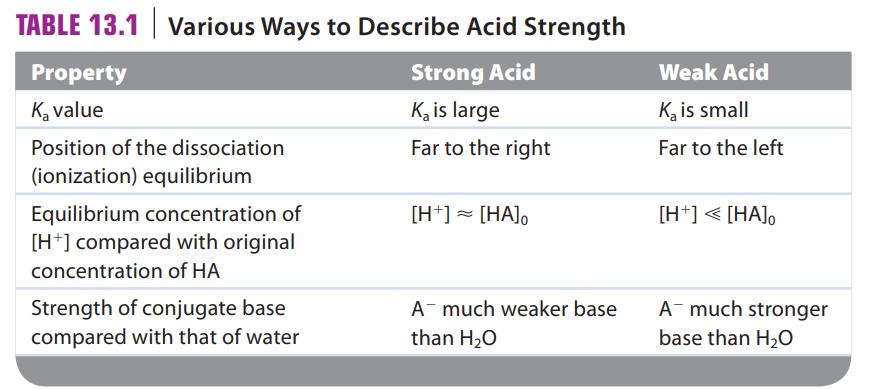
How is acid strength related to the value of Ka? What is the difference between strong acids and weak acids (see Table 13.1)? As the strength of an acid increases, what happens to the strength of the conjugate base? How is base strength related to the value of Kb? As the strength of a base increases, what happens to the strength of the conjugate acid?
Transcribed Image Text:
TABLE 13.1 Various Ways to Describe Acid Strength Strong Acid K₂ is large Far to the right Property K₂ value Position of the dissociation (ionization) equilibrium Equilibrium concentration of [H+] compared with original concentration of HA Strength of conjugate base compared with that of water [H+] = [HA]。 A much weaker base than H₂O Weak Acid K₂ is small Far to the left [H+] < [HA], A much stronger base than H₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
a The acid dissociation constant Ka is a measure of how strongly an acid dissociates in waterThe hig...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Chemists know that nitric and sulfuric acids are strong acids and that acetic acid is a weak acid. They would also agree that ethanol is at best a very weak acid. Acid strength is given directly by...
-
Draw a class diagram that reflect the following Java code segment as shown in Figure 4. public abstract class Vehicle { private int numberofWheels; public Vehicle () { this (0); } protected Vehicle...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Match each of the key terms with the definition that best fits it. ________ The process of ensuring that only authorized changes are made to a system. Here are the key terms from the chapter. The...
-
What would the order of inheritance have been if Ramish had died intestate? In June 2007, Bernard Ramish set up a $48,000 trust fund through West Plains Credit Union to provide tuition for his...
-
You are auditing general cash for Trail Supply Corp. for the fiscal year ended July 31. The client has not prepared the July 31 bank reconciliation. After a brief discussion with the owner, you agree...
-
List and draw the basic flowchart symbols.
-
Refer to Table 55. a. Calculate the ask price of the T-bill maturing on October 3, 2013, as of June 21, 2013. b. Calculate the bid price of the T-bill maturing on December 19, 2013, as of June 21,...
-
Penne Pharmaceuticals sold 16 million shares of its $1 par common stock to provide funds for research and development. If the issue price is $13 per share, what is the journal entry to record the...
-
Although the company has already established medical and retirement plan benefits, Toni Prevosti wants to consider other benefits to attract employees. As the companys accountant, you have been...
-
Define or illustrate the meaning of the following terms: a. Amphoteric b. K w reaction c. K w equilibrium constant d. pH e. pOH f. pK w Give the conditions for a neutral aqueous solution at 25C, in...
-
At 35C, K = 1.6 10 -5 for the reaction If 2.0 moles of NO and 1.0 mole of Cl 2 are placed into a 1.0-L flask, calculate the equilibrium concentrations of all species. 2NOCI(g)2NO(g) + Cl(g)
-
A uniform plate has a weight of 50 lb. Link AB is subjected to a couple moment of M = 10 lb ft and has a clockwise angular velocity of 2 rad/s at the instant = 30. Determine the force developed in...
-
A baseball bat when it hits a baseball exchanges a force of 11,000 N. You can model the elasticity of a baseball as spring with a spring constant of 1.1x10 6 N/m. By how much will the baseball be...
-
Records available for the latest period are as follows: Support Depts. Customer Service Administration 580,000 $ 7 100 Total Costs $ # of Employees # of Customer Inquiries Operating Depts. Shirts...
-
Let f(x) = 8 cos x = 8 cos x 7 tan x f'(x) = = 8(-sin(x)) ()- 3
-
Explain what is an organizational code of ethics (Ch 3 topic)? Why should organizations have a code of ethics? Explain the "three nudge systems" discussed in the media clip. Explain why or why not,...
-
Name the type of shift used to describe the difference in the absorption and emission wavelengths and explain what happens to the energy?
-
Green City Builders' balance sheet data at May 31, 2016, and June 30, 2016, follow: For each of the following situations with regard to common stock and dividends of a corporation, compute the amount...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
Draw the major product that is expected when each of the following compounds is treated with excess methyl iodide followed by aqueous silver oxide and heat: (a) Cyclohexylamine (b)...
-
Propose a synthesis for the following transformation (be sure to count the carbon atoms): Br
-
Compound A is an amine that does not possess a chirality center. Compound A was treated with excess methyl iodide and then heated in the presence of aqueous silver oxide to produce an alkene. The...
-
Blooper Industries must replace its magnoosium purification system. Quick & Dirty Systems sells a relatively cheap purification system for $ 1 2 million. The system will last 4 years. Do - It - Right...
-
What is the second and seventh term of the expended binomial: (a+b)^(7)
-
essay on the United Nations Human Rights Council's 2017 report on the financialization of housing (relevant links below). You may either agree or disagree with the report. Your grade should not...

Study smarter with the SolutionInn App


