Kevlar, used in bulletproof vests, is made by the condensation copolymerization of the monomers Draw the structure
Question:
Kevlar, used in bulletproof vests, is made by the condensation copolymerization of the monomers 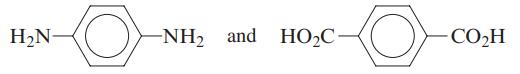
Draw the structure of a portion of the Kevlar chain.
Transcribed Image Text:
H₂N- O -NH₂ and HO₂C CO₂H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
H2N benzene14diami...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2555+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Polyararrud is a term applied ro polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses. a. Kevlar is used in...
-
Polyaramid is a term applied to polyamides containing aromatic groups. These polymers were originally made for use as tire cords but have since found many other uses. a. Kevlar is used in bulletproof...
-
Kevlar is a condensation polymer used in the manufacture of bulletproof vests. Identify the monomers required for the preparation of Kevlar. Kevlar
-
b) Calculate the Fault Level in MVA if a symmetrical bolted 3-phase fault occurs on the LV side of the 5MVA Transformer at 11kV c) The Tranformer impedance is 6% - Calculate the short circuit current.
-
Pierson Inc. operates a retail operation that purchases and sells snowmobiles, amongst other outdoor products. The company purchases all merchandise inventory on credit and uses a perpetual inventory...
-
What are the conditions that make in-house research preferable? What are the conditions that make outside research preferable? Would the company in the opening vignette have been better off to do the...
-
Below is a list of transactions for July 20x6 for Painting Jobs Done Ltd in its first month of operations: Required 1 Record all transactions in the general journal. 2 Prepare a trial balance as at...
-
The inventory, purchases, and sales of Product CAT for March and April are listed below. The company closes its books at the end of each month. It uses the periodic inventory system. Required 1....
-
How much water should be consumed every two hours for a person to run 16 miles? Round your final answer to the nearest whole number. Provide your answer below: ounces
-
The financial statements of Saturn plc have been prepared as follows: Further information: (a) Extract from statement of income 000 Operating profit.........................1,008 Dividend...
-
What kind of decay, if any, would you expect for the nuclei with the energy-level diagrams shown in Figure Q30.16? Energy (a) 22 22- Neutrons FIGURE Q30.16 Protons Energy (b) Neutrons 22 22- Protons...
-
CoCl 4 2- forms a tetrahedral complex ion and Co(CN) 6 3- forms an octahedral complex ion. What is wrong about the following statements concerning each complex ion and the d orbital splitting...
-
The flow rate of ammonia at 10C ( = 624.6 kg/m 3 and = 1.697 10 4 kg/ms) through a 2-cm-diameter pipe is to be measured with a 1.5-cm-diameter flow nozzle equipped with a differential pressure...
-
Explain how demand theory is reflected in the Memorial Health Systems organizational financial statements. Explain how and why market behavior affects the financial statements at various times...
-
Choose any one of the articles from the link below ... read it and in this forum review it for your classmates. Summarize the key points and your views of them. You must use the terms and knowledge...
-
How can leaders cultivate a personal philosophy of ethical leadership, and what strategies can they use to maintain their ethical compass while navigating complex organizational politics and external...
-
Under the current method of production, small heaters pass through several departments, where each department has a collection of similar machines. The first department cuts a metal pipe into one of...
-
A country is experiencing unemployment of 4% and inflation of 10%. The central bank uses contractionary monetary policy to reduce inflation. Explain how this might happen, what policy might be used,...
-
Fred and Emily are among the 50 finalists in a lottery in which 5 winners will be randomly selected to receive a free vacation in Hawaii. Using the hypergeometric distribution, determine the...
-
Solve each equation. x 3 - 6x 2 = -8x
-
Use the geometrical construction shown in Example Problem 24.8 to derive the energy levels of the cycloheptatrienyl cation. What is the total energy of the molecule? How many unpaired electrons will...
-
One of the low-energy geometries of digermane, Ge 2 H 2 , is ethene-like. The Lewis dot structure shown
-
S p hybridization on each Ge atom in planar trans-digermane has been described as sp 1.5 for the GeGe sigma bond and sp 1.8 for the GeH bond. Suppose that the Ge lone electron (in terms of Lewis dot...
-
Cost of Production Report Hana Coffee Company roasts and packs coffee beans. The process begins by placing coffee beans into the Roasting Department. From the Roasting Department, coffee beans are...
-
More info 1. Suppose you invest a sum of $2,000 in an interest-bearing account at the rate of 10% per year. What will the investment be worth six years from now? 2. How much would you need to invest...
-
Schriever Corporation is an oil well service company that measures its output by the number of wells serviced. The company has provided the following fixed and variable cost estimates that it uses...

Study smarter with the SolutionInn App


