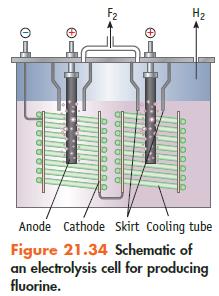
If an electrolytic cell for producing F 2 (Figure 21.34) operates at 5.00 10 3 A
Question:
If an electrolytic cell for producing F2 (Figure 21.34) operates at 5.00 × 103 A (at 10.0 V), what mass of F2 can be produced per 24-hour day? Assume the conversion of F− to F2 is 100%.
Data given in Figure 21.34
Transcribed Image Text:
H₂ Anode Cathode Skirt Cooling tube Figure 21.34 Schematic of an electrolysis cell for producing fluorine.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
According to Faradays law of electrolysis W Ite ...View the full answer

Answered By

Somil Agarwal
I am a student of NISER, BBSR which is the biggest research institute in India after IISc, Banglore.
I am a theoretical Physicist specialized in Condensed Matter.
I know Quantum Mechanics, Many-body Physics, Electronics, Solid State Physics, Classical Mechanics, Numerical Analysis, Linear Algebra, Probability Theory, Differential Equation, Quantum Information and many other topics related to Physics and Mathematics.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
What mass of AgI can be produced from a 0.512-g sample that assays 20.1% AlI3?
-
Elemental calcium is produced by the electrolysis of molten CaCl2. (a) What mass of calcium can be produced by this process if a current of 7.5 103 A is applied for 48 h? Assume that the...
-
The city of Opelika was having a problem ting land for a new sanitary landfill when the Alabama Energy Extension Service offered the solution of burning the solid waste to generate steam. At the same...
-
A machine was sold in December 20x3 for $13,000. It was purchased in January 20x1 for $19,000, and depreciation of $16,000 was recorded from the date of purchase through the date of disposal....
-
First Security Bancorp Inc. wishes to evaluate three capital investment projects by using the net present value method. Relevant data related to the projects are summarized as follows: Instructions...
-
Going Places, Inc., manufactures a variety of luggage for airline passengers. The company has several luggage production divisions, including the Suitable Cases Division, as well as a wholly owned...
-
A comparative balance sheet for Cirrus Corporation is provided in the Working Papers. The income statement for the current year indicates that net income was \($20,900.00\) and the depreciation...
-
At the beginning of last year, Diekow Productions set budgeted fixed overhead costs at $456,000. During the year, actual fixed overhead costs were $500,000. Using this information and the applicable...
-
Bill Pei, CPA, is about to begin his audit of the accuracy of his client's accounts receivable. Based on experience, he expects that approximately 1 percent of the client's 40,000 accounts have...
-
Halogens combine with one another to produce interhalogens such as BrF 3 . Sketch a possible molecular structure for this molecule, and decide if the FBrF bond angles will be less than or greater...
-
To prepare chlorine from chloride ion a strong oxidizing agent is required. The dichromate ion, Cr 2 O 7 2 , is one example (Figure 21.35). Consult the table of standard reduction potentials...
-
Name the two basic projections used in state plane coordinate systems. What are their fundamental differences? Which one is preferred for states whose long dimensions are north-south? East west?
-
Net Realizable Value Method, Decision to Sell at Split-off or Process Further Pacheco, Inc., produces two products, overs and unders, in a single process. The joint costs of this process were...
-
Compare and contrast UAS communication systems in terms of appropriate application, efficacy and reliability.How do you decide which is most appropriate for your application?
-
The maturity value of a savings account that belonged to Peach Company was $41,888.50. The interest charged for the first 5 years was 3% compounded semi-annually, and 4% compounded quarterly for the...
-
Samson Company Case Sampson Company uses a job order cost system with overhead applied to products based on direct labor hours. Based on previous history, the company estimated its total overhead for...
-
(1) Were most of the government bonds held by SVB long-term or short-term bonds? (2) How did that aggravate (or mitigate) the SVB financial challenges? (3). Interest Rate Environment Analysis:...
-
Under what conditions will the returns on investing outside the pension fund be greater than the returns on investing inside the pension fund?
-
CRUZ, INC. Comparative Balance Sheets December 31, 2015 CRUZ, INC. Income Statement For Year Ended December 31, 2015 Required Use the indirect method to prepare the cash provided or used from...
-
Considering the lens in Problem 6.29, determine its focal length and the location of the focal points with respect to its vertices V 1 and V 2 . Data from Prob. 6.29 Figure P.6.29 shows two identical...
-
Figure P.6.29 shows two identical concave spherical mirrors forming a so-called confocal cavity. Show, without first specifying the value of d, that after traversing the cavity two times the system...
-
Starting with the exact expression given by Eq. (5.5), show that Eq. (6.46) results, rather than Eq. (5.8), when the approximations for 0 and i are improved a bit. 1 ( n2Si R l; NSo ) (5.5) li lo
-
The following regression was ran to explore the impact of the number of runners in a marathon (in hundreds) on the number of visits to the medical booth at the race. Model parameters(Booth Visits)...
-
Suppose that the S&P 500, with a beta of 1.0, has an expected return of 13% and T-bills provide a risk-free return of 6%. a. What would be the expected return and beta of portfolios constructed from...
-
can you Compare and contrast the similarities and differences of C corporation and S corporations. using these requirements? -Method of incorporation. -Procedures for electing S corporation status....

Study smarter with the SolutionInn App


