Use the data in Appendix D to calculate the chemical atomic mass of lithium, to two decimal
Question:
Use the data in Appendix D to calculate the chemical atomic mass of lithium, to two decimal places.
Appendix D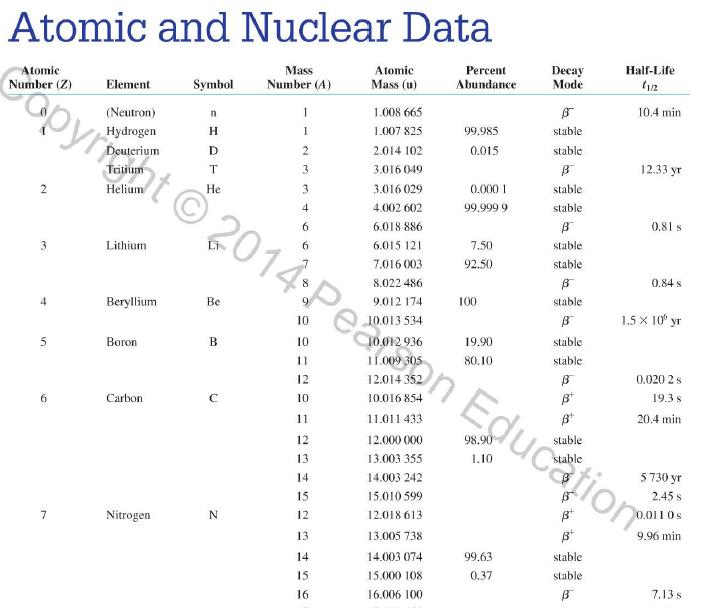
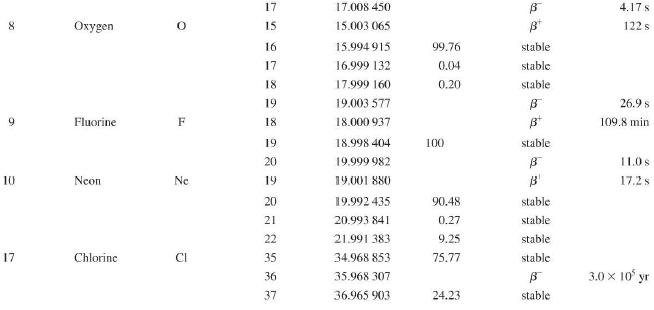
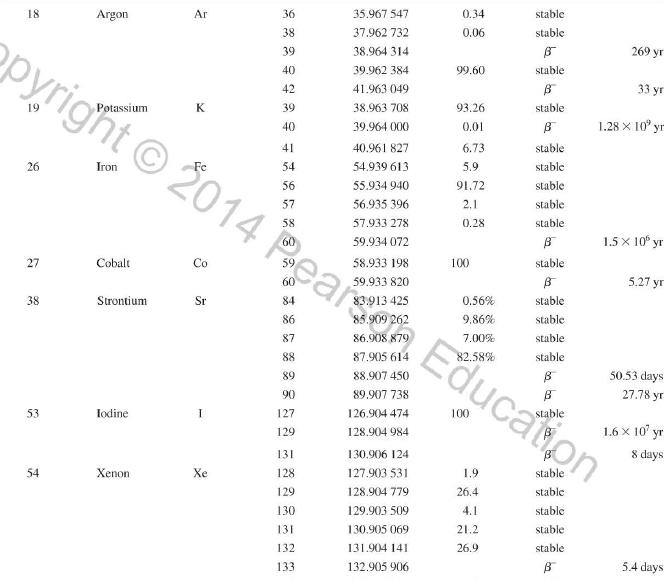
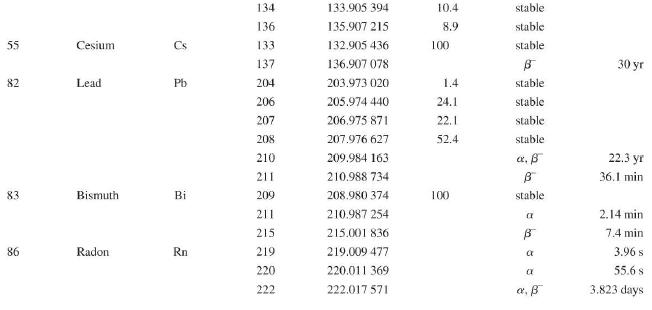
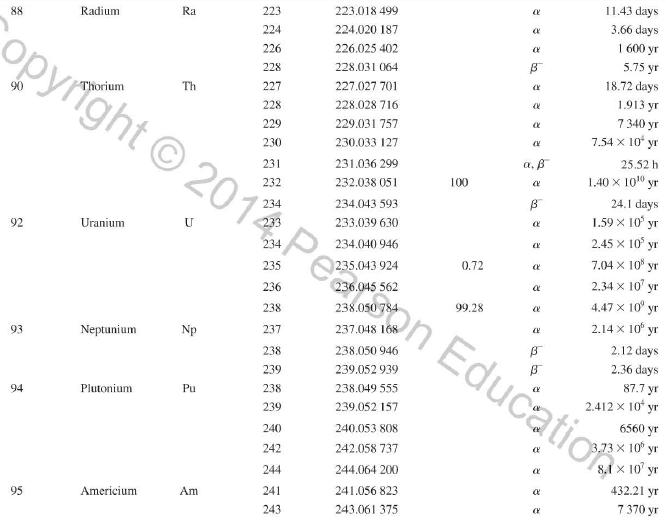
Transcribed Image Text:
Atomic and Nuclear Data Mass Element Symbol Number (A) (Neutron) n H 1 D 2 T 3 3 4 6 3 Lithium Beryllium Be Copyright 2014 Pearson Education. Atomic Mass (u) Percent Abundance Decay Half-Life Mode (1/2 1.008 665 15 10,4 min 1.007 825 99.985 stable 2.014 102 0.015 stable 3.016 049 B 12.33 yr 3.016 029 0.0001 stable 4.002 602 99.999 9 stable 6.018 886 B 0.81 s 6.015 121 7.016 003 7.50 stable 92.50 stable 8.022 486 9.012 174 100 stable 0.84 s B 1.5 x 10" yr 5 Boron B 10 11 12 Carbon C 10 11 11.011 433 B 20.4 min 12 12.000 000 13 13.003 355 14 14.003 242 5 730 yr 15 15.010599 7 Nitrogen N 12 12.018 613 13 13.005 738 B+ 9.96 min 14 14.003 074 99.63 stable 15 15.000 108 0.37 stable 16 16.006 100 B 7.13 s 19.90 stable 80.10 stable B ba ta ba 0.020 2 s 19.3 s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

College Physics A Strategic Approach
ISBN: 9780321907240
3rd Edition
Authors: Randall D. Knight, Brian Jones, Stuart Field
Question Posted:
Students also viewed these Sciences questions
-
Salespeople should think ahead about what questions and statements to include in a conversation and be prepared to hold up their end of the conversation with correct product or service information....
-
Use the data in Appendix D to calculate the chemical atomic mass of neon, to two decimal places. Appendix D Atomic and Nuclear Data Mass Element Symbol Number (A) (Neutron) n H 1 D 2 T 3 3 4 6 3...
-
Suppose we have a linear production function F(L,K)=5L+2K. The prices of labor and capital are w=2 and r=10. a. Prove that this production function exhibits the constant return to scale. b. Find the...
-
Verde Company produces wheels for bicycles. During the year, 657,000 wheels were produced. The actual labor used was 368,000 hours at $9.50 per hour. Verde has the following labor standards: 1)...
-
The following are the financial statements of Nosker Company. NOSKER COMPANY Income Statement For the Year Ended December 31, 2019 Sales revenue.....................................$242,000 Cost of...
-
True or False: 1. In public choice analysis, bureaucrats, politicians, and voters are assumed to make choices that they believe will yield to the public expected marginal benefits greater than their...
-
Ilectronics Inc. makes high-quality speakers for a variety of applications. Electronics Inc. is a St. Louis-based company but has significant manufacturing operations in China. When manufacturing...
-
The Fitzgerald Company maintains a checking account at the Bank of the North. The bank provides a bank statement along with canceled checks on the last day of each month. The October 31, 2007 bank...
-
The following are all part of the Holistic Marketing Concept adopted to ensure cohesive philosophy except A . Relationship marketing B . Integrated marketing C . Green marketing D . Internal marketing
-
238 U is long-lived but ultimately unstable; it will eventually spontaneously break into two fragments, a 4 He nucleus and a 234 Th nucleus, in a process called alpha decay, which we'll learn about...
-
a. Is there a stable \({ }_{3}^{30} \mathrm{Li}\) nucleus? Explain how you made your determination. b. Is there a stable \({ }_{92}^{184} \mathrm{U}\) nucleus? Explain how you made your determination.
-
If M is a k-dimensional manifold with boundary, prove that M is a (k - 1) -dimensional manifold and M - M is a k=dimensional manifold.
-
1. What specific marketing advantages does having pop-ups in Walmart provide for Sleep Country? How do you think these pop-ups will impact Sleep Country's iconic jingle "Why buy a mattress anywhere...
-
To record the purchase of inventories for 150,000 when the exchange rate is $1.30:1. Company purchases forward contract to buy 150,000 at $1.35:1. Exchange rate at maturity is $1.45:1. Required:...
-
The given expression is the limit of a Riemann sum of a function fon [a, b]. Write this expression as a definite integral on [a, b]. 11 5ck lim 2 nk=1c +7 4x, [1,2]
-
1. Given the utility function 1/2 1/3 U = X X au au Determine the value of marginal utilities and at the point (25,8). Hence 1 ????z (a) estimate the change in utility when both x and x increase by 1...
-
Find literature values for room temperature magnetic susceptibility for graphite, manganese zinc ferrite, germanium, and iron. Are they within the same order of magnitude? Why or why not?
-
How is the periodic interest expense affected by the amortization of the premium on bonds payable?
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
They must stay up-to-date on changes in regulations related to accounting and finance, as well as ensure that their organizations comply with these regulations. a. Compliance b. Forecasting c. Cost...
-
Post Malone has adjusted gross income (AGI) of $1,000,000 on his 2022 tax return, filed April 15, 2023. He unintentionally failed to include $100,000 that he received as an Instagram Influencer. How...
-
Asphalt Company had the following information at year end prior to any adjustments: Accounts Receivable $242,560 Allowance for Doubtful Accounts $3,271 (credit balance) Sales $1,645,730 Asphalt...

Study smarter with the SolutionInn App


