Prove that the process shown in Figure 4-9 is impossible if the cylinder contains a monatomic ideal
Question:
Prove that the process shown in Figure 4-9 is impossible if the cylinder contains a monatomic ideal gas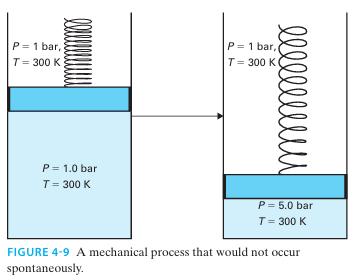
Transcribed Image Text:
P = 1 bar, T= 300 K P = 1.0 bar T = 300 K l l l l l l l l l P= 1 bar, T = 300 K P = 5.0 bar T = 300 K FIGURE 4-9 A mechanical process that would not occur spontaneously.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
A monatomic ideal gas follows the cyclic process shown in the figure. The temperature of the point at the bottom left of the triangle is 470.0 K. (a) How much net work does this engine do per cycle?...
-
A monatomic ideal gas at 27°C undergoes a constant pressure process from A to B and a constant volume process from B to C. Find the total work done during these two processes. 1 L 2L V
-
A monatomic ideal gas at 27°C undergoes a constant volume process from A to B and a constant pressure process from B to C. Find the total work done during these two processes. 2 atm B C 1 atm 1L...
-
Tell whether the given side lengths of a triangle can represent a right triangle. 36, 48, and 60
-
Barnes & Noble sells books, magazines, music, and videos through retail stores and on the Web. For a retailer like Barnes & Noble, inventory is a critical element of the business and it is necessary...
-
Think of the gaudy corporate perks given to managers, such as a plush office, a company jet, or luxury box seats at professional sporting events. How can managers justify these as value-maximizing...
-
The following data are taken from Madison Distribution Company's March payroll: Assume that the company is subject to a 5.2 percent state unemployment tax and an 0.6 percent federal unemployment tax....
-
For the network shown in Figure with all times indicating weeks, answer the following questions: a. What is the impact on the end date of the project if activity B slips by two weeks? b. What is the...
-
1.Name of Center: ABC Child Care Center 2. Ages serviced (youngest to oldest): Infant to 12 Years 3. How many children in total when at capacity? 60 4. How many classrooms will there be? Name each...
-
A stream of liquid nitrogen enters an adiabatic, steady-state valve as a saturated liquid at P = 2 MPa. The material leaves the valve at P = 0.6 MPa. Use the data in Figure 2-3 to determine the...
-
A 10 ounce glass of water (half full) is initially at 15C. It is left outside overnight in a location where the air temperature is 5C. By morning the glass is in equilibrium with the surroundings....
-
Maple Limited (Maple) was incorporated on January 2, Year 1, and commenced active operations immediately in Greece. Common shares were issued on the date of incorporation for 100,000 euros (¬),...
-
Suppose you are deciding whether to locate a factory in China or in Mexico to serve the U.S. market. What factors would influence your location decision? Suppose the price of oil suddenly rises. What...
-
Under what circumstances might public and private partners undertake a strategic alliance?
-
Why does the MFN principle promote multilateral, rather than bilateral, negotiations among WTO members?
-
Assume that you have just been transferred by your company to a new facility in a foreign location. Which of your own personal dimensions do you think will be most effective in helping you deal with...
-
What is international strategic management?
-
There are some dangers in concentrating product-market analysis only upon a firms specific brand and those brands that compete directly with a firms brand. Discuss.
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Grade 1020 steel has a yield strength of 42 ksi and an elastic modulus of 30 Mpsi. Another grade of steel has a yield strength of 132 ksi. What is its elastic modulus?
-
A steel cable of diameter 3/16 in. is attached to an eyebolt and tensioned to 500 lb (Figure P5.4, see on page 226). Calculate the stress in the cable, and express it in the dimensions psi, ksi, Pa,...
-
When a 120-lb woman stands on a snow-covered trail, she sinks slightly into the snow because the compressive stress between her ski boots and the snow is larger than the snow can support without...
-
1. _______________________ is used when you are comparing more than two related groups. a. Factorial ANOVA b. Mixed design ANOVA c. One-way ANOVA d. Repeated measures ANOVA
-
The development costs of perennial plants are reported as a non-current asset on the balance sheet at The development costs of perennial plants are reported as a non-current asset on the balance...
-
Wendy and Wayne are evaluating a project that requires an initial investment of $792,000 in fixed assets. The project will last for fourteen years, and the assets have no salvage value. Assume that...

Study smarter with the SolutionInn App


