Consider a gasifier that receives 4 kmol CO, 3 kmol H 2 , and 3.76 kmol N
Question:
Consider a gasifier that receives 4 kmol CO, 3 kmol H2, and 3.76 kmol N2 and brings the mixture to equilibrium at 900 K, 1 MPa, with the following reaction:
2 CO+ 2H2 ⇔ CH4 + CO2
which is the sum of Eqs. 14.32 and 14.33. If the equilibrium constant is K = 2.679, find the exit flow composition.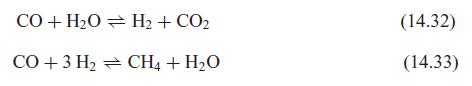
Transcribed Image Text:
CO + H20 = H2 + CO2 (14.32) CO + 3 H2 = CH4 + H20 (14.33)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
To find the exit flow composition we need to calculate the extent of reaction and then use the equil...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Consider an initial mixture of N2 and H2 gases that can be represented as follows. The gases react to form ammonia gas (NH3) as represented by the following concentration profile. a. Label each plot...
-
A mixture of 1 kmol of CO and 2 kmol of O2 is heated to 2000 K at a pressure of 2 atm. Determine the equilibrium composition of (a) O2, (b) CO2 (in kmols) assuming the mixture consists of CO2, CO and...
-
The equilibrium reaction with methane as CH4 C + 2H2 has ln K = -0.3362 at 1440 R and ln K = -4.607 at 1080 R. By noting the relation of K to temperature, show how you would...
-
A group of fraud examiners is coordinating an investigation at a local law firm. Several lawyers at the firm are suspected of overbilling clients, possibly creating fake client accounts, and then...
-
Required Prepare General Journal entries to record the following transactions of Billington Company. 2015 Jan. 10, Accepted a $3,000, 60-day, 6% note dated this day in granting a time extension on...
-
From the e-Activity, select an athlete and study the performance trends of his/her career. Once you have reviewed his/her performance trends, provide three bullets about how his/her career has been...
-
Refer to Exhibits 4-4 and 4-5. Suppose manufacturing costs were the same, but there was an ending work-in-process inventory of $3 million. The cost of the completed goods would therefore be $37...
-
Fogelberg Corporation is a regional company which is an SEC registrant. The corporations securities are thinly traded on NASDAQ (National Association of Securities Dealers Quotes). Fogelberg has...
-
For a cash dividend, the ex-dividend date: Multiple Choice Typically, is one business day before the payment date. Typically, is one business day after the payment date. Typically, is one business...
-
The owner of Neros company has hired you to analyze her company's performance and financial position, as well as the positions of Iits competitors, Centar and Xermix. However, the data Neros abtained...
-
A coal gasifier produces a mixture of 1 CO and 2H 2 that is fed to a catalytic converter to produce methane. This is the methanation reaction in Eq. 14.33 with an equilibrium constant at 600 K of K =...
-
Repeat the previous problem, assuming the argon constitutes 1% of a gas mixture where we neglect any reactions of other gases and find the pressure that will give a mole concentration of A r + of...
-
San Francisco Bay area consumers Wanda Greenwood, Ladelle Hatfield, and Deborah McCleese each signed up for an Aspire Visa credit card marketed by CompuCredit Corp. of Atlanta, Georgia. The card was...
-
Design an SPP that implements the following algorithm. Your design must use the least possible number of states. Your solution should consist of the processor's FSMD, its datapath and the state...
-
Twitter Productions, Inc. has manufactured 10,000 units of product Tweets in September at the following costs: Opening work in process inventory P 44,062.50 Indirect materials used (10% of materials...
-
A company had pretax accounting income of $ 1 , 5 0 0 , 0 0 0 . The company had the following data: Income from exempt municipal bonds: $ 7 0 , 0 0 0 Depreciation for tax purposes in excess of...
-
Performance management systems. Summarize the main discussion points from the articles that you have read and detail the implications for employees. Select three examples of these best practices in...
-
(b) Sodium chloride is an ionic compound with the chemical formula NaCl. (i) Give the electron configurations of sodium (Na) and chlorine (Cl) using spdf notation. [2 marks] (ii) Use electron...
-
Why does the internal rate of return equate to a net present value of zero?
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
When aniline is treated with fuming sulfuric acid, an electrophilic aromatic substitution reaction takes place at the meta position instead of the para position, despite the fact that the amino group...
-
Para-Nitroaniline is an order of magnitude less basic than meta-nitroaniline. (a) Explain the observed difference in basicity. (b) Would you expect the basicity of ortho-nitroaniline to be closer in...
-
Methadone is a powerful analgesic that is used to suppress withdrawal symptoms in the rehabilitation of heroin addicts. Identify the major product that is obtained when methadone is subjected to a...
-
Paste X v Calibri (Body) <11 14 BIU fx v A 10 h Wrap Text Merge & Center C D E F The following data from the just completed year are taken from the accounting records of Fatimah Company (in dollars):...
-
Eisner Company has an opportunity to manufacture and sell a new product for a five-year period. The company estimated the following costs and revenues for the new product: S Cost of new equipment...
-
Novak Pix currently uses a six-year-old molding machine to manufacture silver picture frames. The company paid $94,000 for the machine, which was state of the art at the time of purchase. Although...

Study smarter with the SolutionInn App


