Use Eq. 12.34 to derive an expression for the derivative (T/v)s. What is the general shape of
Question:
Use Eq. 12.34 to derive an expression for the derivative (∂T/∂v)s. What is the general shape of a constant s process curve in a T–v diagram? For an ideal gas, can you say a little more about the shape?
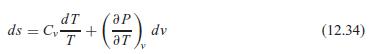
Transcribed Image Text:
IP ds = C,+ dv (12.34) aT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The equation 1234 is the equation of state for an ideal gas given by P RTv Where P ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Use Gauss's law to derive an expression for the electric field outside the thin spherical shell of Conceptual Example 16.8.
-
Use Eq. 13.34 to get an expression for the derivative (T/v)s. What is the general shape of a constant s process curve in a T-v diagram? For an ideal gas can you say a little more about the shape?
-
Derive an expression for the capillary height change h, as shown, for a fluid of surface tension Y and contact angle θ between two parallel plates W apart. Evaluate h for water at 20°C if W...
-
Sentry Transport, Inc., of Atlanta provides in-town parcel delivery services in addition to a full range of passenger services. Sentry engaged in the following activities during the current year: a....
-
What is the difference between managed floating and free floating exchange rates?
-
The following table shows the last six years average new weekly unemployment insurance claims. Year Average weekly new unemployment insurance claims 2004 .............341,300 2005...
-
Ann Keeley and Susie Norton are partners in a business they started two years ago. The partnership agreement states that Keeley should receive a salary allowance of $30,000 and that Norton should...
-
The adjusted trial balance of Business Reduction Systems at March 31, 2016, follows: Requirements 1. Journalize the required closing entries at March 31, 2016. 2. Set up T-accounts for Income...
-
MBA 641 Project 4 Project 4 Objectives: 1. Develop operating budgets 2. Analyze the cash flow of the company. 3. Analyze how changes in cost or revenue impact the budgets. 4. Provide recommendations...
-
Joyce and Marvin run a day care for preschoolers. They are trying to decide what to feed the children for lunches. They would like to keep their costs down, but also need to meet the nutritional...
-
The triple point of carbon dioxide is 56.4C. Predict the saturation pressure at that point using Table B.3.
-
Use Eq. 12.32 to solve for (T/P)s in terms of T, v, C p , and p . How large a temperature change does water at 25C ( p = 2.1 10 4 K1) have when compressed from 100 kPa to 1000 kPa in an isentropic...
-
Of the following four scenarios, which survey results are likely to be the most statistically significant and which are likely to be the least statistically significant? Explain your answer. Scenario...
-
Weston Products manufactures an industrial cleaning compound that goes through three processing departments Grinding, Mixing, and Cooking. All raw materials are introduced at the start of work in the...
-
Review this between the U.S. Deputy Secretary of Defense and the U.S. Secretary of the Air Force. This is an historic exchange that introduces the first attempt by the military to address sexual...
-
Classic Limo, Inc. provides limousine service to Tri-Cities airport. The price of the service is fixed at a flat rate for each trip and most costs of providing the service are stable for each trip....
-
CM Ltd makes a product called Chin. The relevant range of operations is between 2,500 units and 10,000 units of Chin per month. Per unit costs at two activity levels are as follows: 5,000 units at...
-
You are injection molding a part that has parting line dimensions of 200 mm by 250 mm. The machine's clamp is rated to 500 metric tons. Determine the maximum injection pressure possible
-
Which financial ratios are considered to be efficiency ratios?
-
Design a circuit which negative the content of any register and store it in the same register.
-
Identify the reagents you would use to convert 3-pentanone into 3-hexanone.
-
Identify the reagents necessary to achieve each of the following transformations. OEt OEt
-
Draw the structure of the product that is obtained when acetophenone is treated with each of following reagents: (a) Sodium hydroxide and excess iodine followed by H3O+ (b) Bromine in acetic acid (c)...
-
Consider how Star Valley, a popular ski resort, could use capital budgeting to decide whether the $8 million Blizzard Park Lodge expansion would be a good investment. Assur conce assur Click the icon...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Debit Credit Balance 1/1 28,000 Credits Debits 146,000 Balance 12/31 38,000 Manufacturing Overhead...
-
Your company has total receivables outstanding at 31 December 2020 of $20,000. You think that about 2% of these balances will not be collected and wish to make an appropriate allowance. You have not...

Study smarter with the SolutionInn App


