Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to
Question:
Verify that the ideal gas part of the Helmholtz function substituted in Eq. 12.86 does lead to the ideal gas law, as in the note after Eq. 12.97.
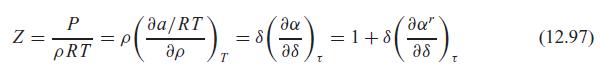
![]()
Transcribed Image Text:
da/RT da" = p pRT ap = 8 as =1+8 a8 (12.97) T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
To verify that the ideal gas part of the Helmholtz function leads to the ideal gas law we can start ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Assuming that the ideal gas law holds, find the amount of nitrogen gas in a container if P = 0.836 atm 0.003 atm, V = 0.01985 m 3 0.00008 m 3 , T = 298.3 K 0.2 K. Find the expected error in the...
-
The ideal gas law relates four variables. An empirical gas law relates two variables, assuming the other two are constant. How many empirical gas laws can be obtained? Give statements of each.
-
The gas law for an ideal gas at absolute temperature T (in kelvins), pressure P (in atmospheres), and volume V (in liters) is PV = nRT, where is the number of moles of the gas and R = 0.0821 is the...
-
A machine fills containers with a particular product. The standard deviation of filling weights computed from past data is 0.6 ounces. If only 2% of the containers hold less than 18 ounces, what is...
-
Suppose 1 = $0.0077 in London, $1 = SF2.00 in New York, and SF1= 65 in Paris. a. If you begin by holding 10,000 yen, how could you make a profit from these exchange rates? b. Find the arbitrage...
-
3 kg of oxygen undergoes a non-flow process during which its pressure remains constant at 7 bar from an initial volume of 1200 litres to a state where the temperature is \(520 \mathrm{~K}\). Find...
-
Great Man leadership theory assumes that: A. All leaders are great men. B. Leadership is determined by genetic and social inheritance. C. Leaders are from the working class D. A and B.
-
On February 17, Walters Corporation acquired 4,000 shares of the 100,000 outstanding shares of Lycore Co. common stock at $22.50 plus commission charges of $200. On July 11, a cash dividend of $0.80...
-
TB Problem Qu. 8-216 Capes Corporation is a wholesaler of ... Capes Corporation is a wholesaler of industrial goods. Data regarding the store's operations follow. Sales are budgeted at $430,000 for...
-
It costs thousands of dollars to generate an application (apps) for the Apple iPad. Just offering an APP is not a guarantee of success. The most successful APPS, sold as downloads, have to offer true...
-
R-410a is a 1:1 mass ratio mixture of R-32 and R-125. Find the specific volume at 20C, 1200 kPa, using Kays rule and the generalized charts, and compare it to the solution using Table B.4.
-
Gases like argon and neon have constant specific heats. Develop an expression for the ideal gas contribution to the Helmholtz function in Eq. 12.92 for these cases. a* = h* RT Ts* (12.92)
-
Suppose that a random sample of fifteen 18- to 34-year-olds living with their parents is selected and asked if they contribute to household expenses. a. Is the selection of the fifteen 18- to...
-
Which cultural dimension causes the most conflict in your friendships and/or family relationships? Explain how the cultural dimension works as though you were explaining it to someone who has never...
-
An air column 8 2 . 5 cm in length is open at both ends. It is resonating at the fourth harmonic. What is the frequency of vibration?
-
sExplore the relationship between deadlock and resource management policies. How can proper resource management help in reducing the likelihood of deadlock in large-scale systems?
-
What do you do to pass the time?In environments like our current one, where more and more people are being mandated to stay at home, what are you solutions for working from home, continuing your...
-
My employer is trying to coerce me into signing an agreement on a new pay structure, which is a reduction of $50,000 from my current pay - a commission- based plan with earning of $250,000. Am I...
-
Find the descriptive measures for 1, 2, 3, 4, 5, 6, 7, 1, 2, 3, 4, 5, 6, 7. a. Obtain the quartiles. b. Determine the interquartile range. c. Find the five-number summary.
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Rank the following compounds in terms of increasing basicity: N. Br z-
-
When (E)-4-amino-3-buten-2-one is treated with molecular hydrogen in the presence of platinum, the resulting amine is more basic than the reactant. Draw the reactant and the product, and explain why...
-
For each of the following compounds, draw the form that predominates at physiological pH: (a) (b) (c) CI CH CI Sertraline (Zoloft) An antidepressant NH2 Amantadine Used in the treatment of...
-
A ( n ) 1 9 - year bond has a coupon of 9 % and is priced to yield 1 4 % . Calculate the price per $ 1 , 0 0 0 par value using semi - annual compounding. If an investor purchases this bond two months...
-
You have to choose between project A and Project B based on the following data.Which one will you select and why? Project AProject B NPV:$10,000$9,000 IRR:10%12%
-
The number of bank branches continues to increase because most customers choose a bank based on convenient location of a branch. states have eased bank branching restrictions since the financial...

Study smarter with the SolutionInn App


