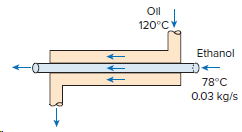
Ethanol is vaporized at 78°C (h fg = 846 kJ/kg) in a double-pipe parallel-flow heat exchanger at
Question:
(a) The LMTD method
(b) The ε-NTU method.
Transcribed Image Text:
Oil 120°C Oil Ethanol 78°C 0.03 kg/s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Ethanol is vaporized by hot oil in a doublepipe parallelflow heat exchanger The outlet temperature a...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Fundamentals of Thermal-Fluid Sciences
ISBN: 978-0078027680
5th edition
Authors: Yunus A. Cengel, Robert H. Turner, John M. Cimbala
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Physics questions
-
The enthalpy of vaporization of ethanol is 38.7 kJ/ mol at its boiling point (78oC). Determine Ssys, Ssurr, and Suniv when 1.00 mole of ethanol is vaporized at 78oC and 1.00 atm.
-
Consider the flow of engine oil (c p = 2048 J/kg·K) through a thin-walled copper tube at a rate of 0.3 kg/s. The engine oil that enters the copper tube at an inlet temperature of 80°C is...
-
Determine the heat transfer area requirements of Problem 8.41 if a 1-2 shell and tube, an unmixed crossflow, and a parallel flow heat exchanger are used, respectively. From Problem 8.41: A...
-
In designing an experiment, the researcher can often choose many different levels of the various factors in order to try to find the best combination at which to operate. As an illustration, suppose...
-
Solve Problem 5.90 if the gate weighs 1000 lb. PROBLEM 5.90 A 4 00d7x 2-ft gate is hinged at A and is held in position by rod CD. End D rests against a spring whose constant is 828 lb/ft. The spring...
-
In 2012 Craig Gonzales opened Craigs Pets, a small retail shop selling pet supplies. On December 31, 2012, Craigs accounting records showed the following: Inventory on December 31, 2012 .... $ 10,100...
-
Teddy Sargent opened an accounting firm on May 1, 2010. During the month of May, the business completed the following transactions: Requirements 1. Open, or set up, the following T-accounts: Cash,...
-
Reformulate the following balance sheet and income statement for a manufacturing concern. Amounts are in millions. The firm bears a 36 percent statutory taxrate. Balance Sheet Assets Operating cash...
-
. Project Information You estimate the investment required to undertake the project will be equal to 20% of the Property Plant and Equipment (PPE) shown in the Core Lithium Ltd 2023 Balance Sheet...
-
You and your spouse are in good health and have reasonably secure careers. You make about $35,000 annually and have opted for life insurance coverage of 3 times your salary through your employer....
-
Water (c p = 4180 J/kgK) enters the 2.5-cminternal-diameter tube of a double-pipe counter-flow heat exchanger at 17C at a rate of 1.8 kg/s. Water is heated by steam condensing at 120C (h fg = 2203...
-
Saturated water vapor at 100°C condenses in a 1-shell and 2-tube heat exchanger with a surface area of 0.5 m 2 and an overall heat transfer coefficient of 2000 W/m 2 ·K. Cold water (c pc =...
-
Suppose a and b are unit vectors in R2. Show that the distance from a to the line through b is the same as the distance from b to the line through a. Use a picture to explain why this holds. How is...
-
Discuss the role of chaperone proteins in maintaining protein homeostasis and describe what happens when these systems fail, leading to diseases like Alzheimer's or Huntington's.
-
How did the Phillips Curve look differently between pre-pandemic period vs. after-pandemic (recovery) period? What were the possible explanations?
-
One ball is dropped from a 4.2-m high balcony at the exact same time a ball is launched straight up with a velocity of 5.2 m/s. At what height do they collide?
-
Compute the cost assigned to ending inventory using ( a ) FIFO, ( b ) LIFO, ( c ) weighted average, and ( d ) specific identification. For specific identification, units sold include 8 5 units from...
-
Dept. Goal - Department employees will provide tutoring or Stem classes at local schools for a total of 100 hours over the next year. Tactical steps: Connect with local schools to gather information:...
-
(a) Show how you can construct a T flip-flop using a J-K flip-flop. (b) Show how you can construct a J-K flip-flop using a D flip-flop and gates.
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
Instruments located at O are part of the ground traffic control system for a major airport. At a certain instant during the takeoff roll of the aircraft P, the sensors indicate the angle = 50 and...
-
In addition to the information supplied in the previous problem, the sensors at O indicate that r = 14 ft/sec 2 . Determine the corresponding acceleration a of the aircraft and the value of .
-
At the bottom of a loop in the vertical (r-) plane at an altitude of 400 m, the airplane P has a horizontal velocity of 600 km / h and no horizontal acceleration. The radius of curvature of the loop...
-
What are three key challenges you face as a leader in business today and how do you currently maneuver to lessen the impact or to negate the impact of these challenges? What one thing must a leader...
-
While technology offers distinct advantages, some associations have encountered challenges with its application, especially on how an association interacts with its members.What are some of the...
-
You are a consultant at a major investment institution. Your institution has a long-standing business relationship with Humberside plc, which is listed on the London Stock Exchange. A new CEO has...

Study smarter with the SolutionInn App


