A possible reaction for converting methanol to ethanol is (a) Calculate r H r S,
Question:
A possible reaction for converting methanol to ethanol is
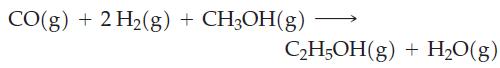
(a) Calculate ΔrH° ΔrS°, and ΔrG° for this reaction at 25 °C.
(b) Is this reaction thermodynamically favored at high or low temperatures? At high or low pressures? Explain.
(c) Estimate K for the reaction at 750 K.
Transcribed Image Text:
CO(g)+ 2 H₂(g) + CH3OH(g) C₂H5OH(g) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a rH fHproducts fHreactants rH 2351 kJmol 2418 kJmol 2007 kJmol 1105 ...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction to prepare methanol from carbon monoxide and hydrogen is exothermic. If you wanted to use this reaction to produce methanol commercially, would high or low temperatures favor a maximum...
-
The vortex tube (also known as a Ranque or Hirsch tube) is a device that produces a refrigeration effect by expanding pressurized gas such as air in a tube (instead of a turbine as in the reversed...
-
Methanol (CH3OH) can be made by the reaction of CO with H2: (a) Use thermochemical data in Appendix C to calculate (Ho for this reaction. (b) To maximize the equilibrium yield of methanol, would you...
-
Casrnir proposes a model of third-culture building to account for the evolutionary nature of intercultural dialogue. What role do you think the many studies on cultural dimensions can play in the...
-
Donnegal Company makes and sells artistic frames for pictures. The controller is responsible for preparing the master budget and has accumulated the following information for 2014. Donnegal has a...
-
How fast, in kilometers per hour, must a sound source be moving toward you to make the observed frequency greater than the true frequency? (Assume that the speed of sound is 340 m/s.)
-
Mr. Lomax owns the mineral rights in some land in Texas. He leases the land to Frank Oil Company, reserving a 1/5 royalty. During 2011, Frank Oil Company makes the following assignments: a. To Mr....
-
Selected transactions for Arnett Advertising Company, Inc., are listed here. 1. Issued common stock to investors in exchange for cash received from investors. 2. Paid monthly rent. 3. Received cash...
-
M Homepage - Northland... M Homepage - Northland... M Homepage - Northla 28. DETAILS Match the trigonometric function with one of the graphs I-VI. f(x) = sec 2x ---Select-- 9 I y II 0 x 3TT
-
Establish at 298 K for the reaction: (a) r S; (b) r H; (c) r G; (d) K. 2 NaHCO3(s) NaCO3(s) + HO(1) + CO(g)
-
Comment on the difficulties of solving environmental pollution problems from the standpoint of entropy changes associated with the formation of pollutants and with their removal from the environment.
-
By using blue paint, determine which of the following FAs accept any words: a. b (1 a. h h
-
Write object-oriented Swift code for the following. The unit codes of four IT units are passed into a method of a class. The units passed in are as follows (and they are passed in this order too):...
-
Guchi and Pluto are in a statistics class. At the end of the semester, after 5 exams, Guchi has a mean of 60 and standard deviation of 20 for exams. Pluto has a mean of 60 and standard deviation of 5...
-
How important is innovation in the Vernon model? I know that the timing of innovation is important, because is has implications on the location of production. Is there another part in which...
-
The pretax financial income (or loss) figures for Sunland Company are as follows. 2015$176,000 2016 243,000 201779,000 2018(176,000) 2019(390,000) 2020 118,000 202190,000 Pretax financial income (or...
-
the account in the ledger of monroe entertainment co . are listed . All accounts have normal balances. account payable 4 3 9 , fees earned 3 2 8 3 , accounts receivable 6 5 7 , insurance expense 5 8...
-
(a) If a proton is accelerated from rest by a potential difference of 1 million volts, how much kinetic energy does it gain? (b) How would your answer to part (a) change if the accelerated particle...
-
Would you use the adjacency matrix structure or the adjacency list structure in each of the following cases? Justify your choice. a. The graph has 10,000 vertices and 20,000 edges, and it is...
-
Standard-costing with beginning and ending work in process. Paquitas Pearls Company (PPC) is a manufacturer of knock off jewelry. Paquita attends Fashion Week in New York City every September and...
-
Operation costing (chapter appendix). Farkas Shoes, a high-end shoe manufacturer, produces two lines of shoes for women. The shoes are identical in design, but differ in the materials used and the...
-
Economic order quantity for retailer. Fan Base (FB) operates a megastore featuring sports merchandise. It uses an EOQ decision model to make inventory decisions. It is now considering inventory...
-
Ariana and John, who file a joint return, have two dependent children, Kai and Angel. Kai is a freshman at State University and Angel is working on her graduate degree. The couple paid qualified...
-
The Foundational 15 [LO6-1, LO6-3, LO6-4, LO6-5, LO6-6, LO6-7, LO6-8] [The following information applies to the questions displayed below.] Oslo Company prepared the following contribution format...
-
Direct Materials Purchases Budget FlashKick Company manufactures and sells soccer balls for teams of children in elementary and high school. FlashKick's best-selling lines are the practice ball line...

Study smarter with the SolutionInn App


