Discuss the following set of rate constants (k) and activation parameters for water exchange reactions of metal
Question:
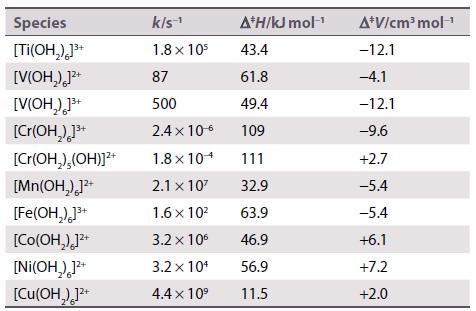
Discuss the following set of rate constants (k) and activation parameters for water exchange reactions of metal aqua ions.
Transcribed Image Text:
Species [TI(OH₂)]³+ [V(OH₂)]²+ [V(OH₂)]³+ [Cr(OH₂)]³+ [Cr(OH₂),(OH)]²+ [Mn(OH₂)]²+ [Fe(OH₂)]³+ [Co(OH₂)]²+ [Ni(OH₂)]²+ [Cu(OH)]²+ k/s-¹ 1.8 x 105 87 500 2.4 x 10-6 1.8 x 104 2.1 x 10² 1.6 x 10² 3.2 x 10⁰ 3.2 x 10¹ 4.4 x 10⁹ A*H/kJ mol-¹ 43.4 61.8 49.4 109 111 32.9 63.9 46.9 56.9 11.5 A*V/cm³ mol-¹ -12.1 -4.1 -12.1 -9.6 +2.7 -5.4 -5.4 +6.1 +7.2 +2.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The set of rate constants k and activation parameters for water exchange reactions of metal aqua ions provides valuable insights into the kinetics and mechanisms of these reactions Lets discuss the si...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
As a specific biological example for Exercise 24.2 and Fig. E24.2(b), the synthesis of tryptophan can be described by the following set of material balances: where k 1 , k 2 , k 3 , and k 4 represent...
-
The following set of questions relates to using Poisson regression methods to analyze data from an in vitro study of human chromosome damage. In this study, using Poisson regression is appropriate...
-
The following set of data shows the number of alcoholic drinks that students at a Kansas university reported they had consumed in the past month: a. Assume the data set is a sample. Calculate the...
-
A U.S Company David Inc. sold merchandise to Fernando SA for 10,000 on September 14, 2017. The spot rate for euro is $0.720 on this day. On October 12, 2018, David Inc. purchased merchandise from...
-
At the end of Roshek Department Stores fiscal year on December 31, 2014, these accounts appeared in its adjusted trial balance. Freight-In ................$ 5,600 Inventory ................ 40,500...
-
Which of the following is not a characteristic of the factor-weighting approach to supplier evalution? a) It applies quantitative scores to qualitative criteria b) The weights typically sum to 100%...
-
Horizontal analysis analyzes: a. Comparable companies. b. Changes in expenses as a percent of sales. c. Changes in expenses as a percent of total assets. d. Changes in balances from one year to...
-
Last year (2013), Simmons Company installed new factory equipment. The owner of the company, Gene Simmons, recently returned from an industry equipment exhibition where he watched computerized...
-
In a manufacturing company, production and inventory both take place. Class, could you explain the relationship between the acquisition and payment cycle and the inventory and warehousing cycle in...
-
Use the concept particularly the effects of penetration and shielding on the radial wavefunction, to account for the variation of single-bond covalent radii with position in the periodic table.
-
Given the following mechanism for the formation of a chelate complex, derive the rate law for the formation of the chelate. Discuss the step that is different from that for two monodentate ligands....
-
The workbook named Quarterlygnpdata.xlsx contains quarterly GNP data for the United States (in billions of 1996 dollars). Extract this data to three separate columns in which the first column...
-
Prepare the statement ot cash flows for the current year ended December 3 1 , using the indirect method to report cash flows from operating activities. Indirect Method- Preparing a Statement of Cash...
-
Current Year 1 Year Ago 2 Years Ago $ 35,677 64,366 85,055 $ 31,780 91,197 111,223 Simon Company's year-end balance sheets follow. At December 31 Assets Cash Accounts receivable, net Merchandise...
-
- hiTiIT a. Venedict invested $86,000 cash along with office equipment valued at $21,000 in a new business named HV Consulting in exchange for common stock. b. The company purchased land valued at...
-
Answer these three questions about the Walmart case study. Must be 1.5-2 pages in length. It is easy to become enamored with a company when it has enjoyed so much success over the years. What are...
-
Find f-1 (x) for f (x) = 15 + 12x.
-
Voluntary environmental programs were extremely popular in the United States, Europe, and Japan in the 1990s. Part of their popularity stems from the fact that these programs do not require...
-
Medi-Exam Health Services, Inc. (MEHS), located in a major metropolitan area, provides annual physical screening examinations, including a routine physical, EKG, and blood and urine tests. MEUS's...
-
Predict the stereochemical outcome of radical bromination of the following alkanes: (a) (b) (c) (d)
-
Compound A has molecular formula C 5 H 11 Br. When compound A is treated with bromine in the presence of UV light, the major product is 2,2-dibromopentane. Treatmentof compound A with NaSH (a strong...
-
In calculating H o R at 285.15 K, only the H o f of the compounds that take part in the reactions listed in Tables 4.1 and 4.2 (Appendix B, Data Tables) are needed. Is this statement also true if you...
-
Using the balance sheet for MNO Corp. as of December 31, 2023, evaluate the companys financial leverage and efficiency. MNO Corp. Balance Sheet As of December 31, 2023 Assets Current Assets Cash...
-
Scenario 2 A 60-kg subject rides a Monark cycle ergometer for 30 min at a cadence of 80 rpm with a mass of 1.5 kg in the basket. The flywheel moves 6 m with every turn of the pedal. Remember that...
-
5. A local factory would like to install a new piece of equipment and would like for you to use your crane (on tracks) to transfer the equipment from a truck into a new building. The equipment weighs...

Study smarter with the SolutionInn App


