Figure 21.24 (which is based on J.B. Goddard and F. Basolo, Inorg. Chem., 1968, 7, 936) shows
Question:
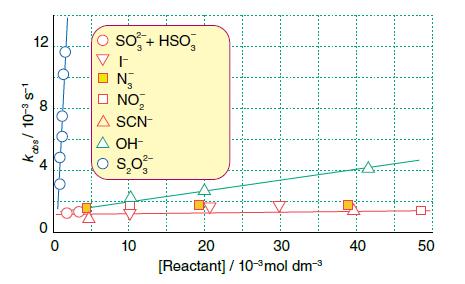
Figure 21.24 (which is based on J.B. Goddard and F. Basolo, Inorg. Chem., 1968, 7, 936) shows the observed first-order rate constants for the reaction of [PdBrL]+ with various Y− to give [PdYL]+, where L is Et2NCH2CH2NHCH2CH2NEt2. Note the large slope for S2O32− and zero slopes for Y− = N3− , I− , NO2−, and SCN−. Propose a mechanism.
Figure 21.24.
Transcribed Image Text:
/ 10-³ S-1 12 8 لمهم 0 0 SO² + HSO 7 F N₂ □NO₂ A SCN- A OH- O S₂0² 04 10 20 30 [Reactant]/10-mol dm-³ 40 50
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
1 Large Slope for S 2 O 3 2 The large slope for S 2 O 3 2 suggests that this species is likely invol...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The following set of questions relates to using Poisson regression methods to analyze data from an in vitro study of human chromosome damage. In this study, using Poisson regression is appropriate...
-
Start of Payroll Project 7-3a October 9, 20-- No. 1 The first payroll in October covered the two workweeks that ended on September 26 and October 3. This payroll transaction has been entered for you...
-
Esther earned 528.00 for the week. If she saved 128.00, how many percent of her earnings for the week did she save?
-
Wiggins Company has net sales of $400,000 and cost of goods available for sale of $300,000. If the gross profit rate is 35%, what is the estimated cost of the ending inventory ? Show computations.
-
The type of chart used to control the central tendency of variables with continuous dimensions is: a) x-chart. b) R-chart. c) P-chart. d) C-chart. e) None of the above.
-
Points quotations In Table 3.16, consider the spot quotations in dollars per pound (\($/)\) and the forward quotations in basis points (1/10,000). On the basis of the points quotations in Table 3.16,...
-
Deglman Manufacturing uses a job order cost system and applies overhead to production on the basis of direct labor costs. On January 1, 2012, Job No. 50 was the only job in process. The costs...
-
With the end of Lockdown due to Covid19, Facebook buys $395,380 for real estate with land, land improvements, and a building. Their goal is to get everyone back to the office. The land is appraised...
-
Octahedral complexes of metal centres with high oxidation numbers or of d metals of the second and third series are less labile than those of low oxidation number and d metals of the first series of...
-
Solutions of [PtH 2 (PMe 3 ) 2 ] exist as a mixture of cis and trans isomers. Addition of excess PMe 3 led to formation of [PtH 2 (PMe 3 ) 3 ] at a concentration that could be detected using NMR....
-
Describe and contrast the role of RFDC and RFID for logistics and supply chain applications.
-
a. Plot the series. What can you learn from examining this plot? b. Calculate and display the first 24 autocorrelations for the series. What do the ACF and PACF suggest about the series? c. Suggest a...
-
If you deposit $400 each year for 8 years in an account that pays 7%, How much will you have at the end of the 8 years? Use the TVM keys on your calculator... NOT the tables in the book Carry answer...
-
3) Find these terms of the sequence {an}, where an -2 (-3)" +5". a) ao b) a c) 24 d) as Answer: a) 3 b) c) d)
-
On January 2 of the current year, BC Co. acquired 1,000 shares of its $10 par value common stock for $20,000. On July 1, BC exchanged this stock for land to be held for use as the site of the...
-
A firm has three categories of budgeted overhead costs. Supervisor Salaries: $ 7 5 0 , 0 0 0 Equipment Lease: $ 1 9 0 , 0 0 0 Maintenance: $ 7 5 , 0 0 0 After an activity - based costing study, the...
-
The accompanying table presents data from the U.S. Census Bureau on median and mean income of male workers for the years 1972 and 2009. The income figures are adjusted to eliminate the effect of...
-
Outline a general process applicable to most control situations. Using this, explain how you would develop a system to control home delivery staff at a local pizza shop.
-
Compound A has molecular formula C 7 H 12 . Hydrogenation of compound A produces 2 methylhexane. Hydroboration-oxidation of compound A produces an aldehyde. Draw the structure of compound A, and draw...
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) Br Br
-
1,2-Dichloropentane reacts with excess sodium amide in liquid ammonia to produce compound X. Compound X undergoes acid-catalyzed hydration to produce a ketone. Draw the structure of the ketone...
-
Finite element formulations in two dimensions can only be constructed as a mappings from a bi-unit square domain. False True
-
Online Driver Education Course with 6 Hours of Training Chapter 12 QUIZ Question 1 to 10 1. Always look carefully for motorcycles before you change lanes because: Their smaller size makes them harder...
-
Consider the price - taker market for dress shirts. The following graph shows the marginal cost ( MC MC ) , average total cost ( ATC ATC ) , and average variable cost ( AVC AVC ) curves for a typical...

Study smarter with the SolutionInn App


