Refer to Table 21.13 and calculate the rate constants for electron transfer in the oxidation of by
Question:
Refer to Table 21.13 and calculate the rate constants for electron transfer in the oxidation of
![]()
by the oxidants
(a)
![]()
(b)
![]()
Comment on the relative sizes of the rate constants.
Table 21.13.
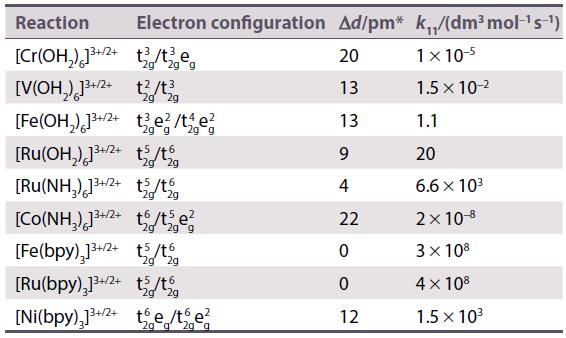
Transcribed Image Text:
[V(OH₂) 1²+ (E° (V³+/V²+) = -0.255V)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Based on the information provided in Table 2113 we can calculate the rate constants for electron tra...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Inorganic Chemistry
ISBN: 9780198768128
7th Edition
Authors: Mark Weller, Tina Overton, Jonathan Rourke
Question Posted:
Students also viewed these Sciences questions
-
Hansel and Gretel are twins. Gretel goes to a star which is 65 light years away with a constant velocity plane (= v/c) = 0,88. Every birthday, the two do not forget to send each other radio signals,...
-
The photosynthetic reaction centre of the purple photosynthetic bacterium Rhodopseudomonas viridis contains a number of bound cofactors that participate in electron transfer reactions. The following...
-
The equilibrium Ac=' B + C at 25C is subjected to a temperature jump that slightly increases the concentrations of Band C. The measured relaxation time is 3.0 us. The equilibrium constant for the...
-
Define price and name the various types of prices described in this chapter.
-
Ryder Company's balance sheet shows Inventory $162,800. What additional disclosures should be made?
-
Altman Distributors, Inc., fills catalog orders. Samples of size n = 100 orders have been taken each day over the past 6 weeks. The average defect rate was .05. Determine the upper and lower limits...
-
The bidask spread (I) In Table 3.14, consider the bid and ask spot quotations in dollars per pound (\($/),\) i.e. in American terms: a. Compute the bid and quotations in European terms, i.e. in...
-
Par, Inc., produces a standard golf bag and a deluxe golf bag on a weekly basis. Each golf bag requires time for cutting and dyeing and time for sewing and finishing, as shown in the following table:...
-
QUESTION 3 (15 MARKS) Lump Sum Purchase of Plant Assets With the end of Lockdown due to Covid19, Facebook buys $395,380 for real estate with land, land improvements, and a building. Their goal is to...
-
Solutions of [PtH 2 (PMe 3 ) 2 ] exist as a mixture of cis and trans isomers. Addition of excess PMe 3 led to formation of [PtH 2 (PMe 3 ) 3 ] at a concentration that could be detected using NMR....
-
Write the rate law for formation of [MnX(OH 2 ) 5 ] + from the aqua ion and X . How would you determine if the reaction is d or a?
-
Suppose fY (y) =4y3, 0 y 1. Find P (0Y 1/2).
-
Sean threw a football to alexander. The ball left seans hand at a height of 2 meters. The ball reached its maximum height 2 seconds after sean released it and hit the ground 3 seconds later....
-
The Fancy Umbrella Company makes beach umbrellas. The production process requires 3 square meters of plastic sheeting and a metal pole. The plastic sheeting costs $0.50 per square meter and each...
-
Terrell Trucking Company is in the process of setting its target capital structure. The CFO believes that the optimal debt-to-capital ratio is somewhere between 20% and 50%, and her staff has...
-
A negative charge of -0.590 C exerts an upward 0.700-N force on an unknown charge that is located 0.400 m directly below the first charge. Part A What is the value of the unknown charge (magnitude...
-
Consider an economy with two individuals, Ann and Bob and two goods x and y. Ann's initial endowment is (50, 20) and Bob's initial endowment is (20,50). Ann's utility function is u(x,y)=xy and Bob's...
-
The accompanying table contains data on the U.S. economy for the years 1983 and 2010. The second column shows the poverty threshold. The third column shows the consumer price index (CPI), a measure...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Give an example of two systems separated by a wall that are in thermal but not chemical equilibrium.
-
A 1.50 mole sample of an ideal gas at 28.5C expands isothermally from an initial volume of 22.5 dm 3 to a final volume of 75.5 dm 3 . Calculate w for this process a. For expansion against a constant...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Polypropylene (PP) is a hydrocarbon polymer produced from the reaction of chemicals obtained from crude oil. PP is used in many applications including the manufacture of food containers such as...
-
Roger lost his job at the R - gone Manufacturing Company. Since then he has been trying to find a job at other local factories. He is counted as employed unemployed not in the labor force
-
Research by Sebastien Bradley of Drexel University and Naomi Feldman of the Federal Reserve revealed that the price elasticity of demand for airline tickets increased after airlines had to reveal tax...

Study smarter with the SolutionInn App


