Find the (lowest-order) relativistic correction to the energy levels of the one-dimensional harmonic oscillator. Use the technique
Question:
Find the (lowest-order) relativistic correction to the energy levels of the one-dimensional harmonic oscillator. Use the technique of Problem 2.12.
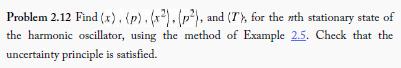
Transcribed Image Text:
Problem 2.12 Find (x). (p). (x²). (p²), and (T), for the nth stationary state of the harmonic oscillator, using the method of Example 2.5. Check that the uncertainty principle is satisfied.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Equation 755 E E E 1 2mc But Problem 212 2mc 2 x w 2 n ...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Quantum Mechanics
ISBN: 9781107189638
3rd Edition
Authors: David J. Griffiths, Darrell F. Schroeter
Question Posted:
Students also viewed these Engineering questions
-
The velocity vs. time graph of an object is shown to the right. Determine the total distance traveled by the object. Show your work. Distance = (m) 20 6 Time (s) 10 12
-
The ground-state energy of a harmonic oscillator is 5.60eV. If the oscillator undergoes a transition from its n = 3 to n = 2 level by emitting a photon, what is the wavelength of the photon?
-
The lowest four energy levels for atomic vanadium (V) have the following energies and degeneracies: What is the contribution to the average energy from electronic degrees of freedom for V when T =...
-
Find a harmonic function (x, y) in the right-half plane x > -1 such that on the boundary (-1, y) = 0, y
-
Refer to the previous exercise. Suppose z = 2.50 instead of 1.04. a. Find the P-value for (i) Ha: p > 0.50. (ii) Ha: p 0.50, and (iii) Ha: p < 0.50. b. Do any of the P-values in part a provide...
-
Ozone gas (O 3 , solute A) dissolved in high-purity water is commonly used in wet cleaning processes associated with semiconductor device fabrication. It is desired to produce a liquid water stream...
-
If accounts receivables increase by \($15,000\) and accounts payables increase by \($11,000,\) the effect on cash flow is a. Increase of $26,000 b. Increase of $4,000 c. Decrease of $4,000 d....
-
a. You created an interactive StaffDinnerEvent class that obtains all the data for a dinner event for Carlys Catering, including details about the event and all the staff members required to work at...
-
Many firms use on-the-job training to teach their employees computer programming. Suppose you work in the personnel department of a firm that just finished training a group of its employees to...
-
Using a linear programming framework and the data from case Exhibit 1, formulate and solve the problem of finding the cost-minimizing set of bids that meet Mars' business requirements and determines...
-
Show that P 2 is hermitian, for hydrogen states with = 0. For such states is independent of and , so (Equation 4.13). Using integration by parts, show that Check that the boundary term vanishes for...
-
(a) Express the Bohr energies in terms of the fine structure constant and the rest energy (mc 2 ) of the electron. (b) Calculate the fine structure constant from first principles (i.e., without...
-
Mr. Husker's Tuxedos Corp. ended the year 2018 with an average collection period of 32 days. The firm's credit sales for 2018 were $56.1 million. What is the year-end 2018 balance in accounts...
-
Would you consider YSNs founder a transformative or a transactional leader? What kind of leader is called for in YSNs situation? Given a choice, with which type of leader would you prefer to work?...
-
A social agency based in a midwestern city that provides mental health services has an annual budget of $80 million, funded almost exclusively by government contracts. Is an organization of this...
-
The Filer Commissions recommendation to establish a permanent agency on charitable nonprofits within the Treasury Department was not implemented. If you could create such an agency now, what would be...
-
From the information in the first section of the case study, how would you characterize YSNs organizational culture? Do you have enough information to have a sense of the deepest level of its...
-
Is organizational culture shaped by the external environment or vice versa? On what do you base your views?
-
As its year-end approaches, it appears that Mendez Corporation's net income will increase 10% this year. The president of Mendez Corporation, nervous that the stockholders might expect the company to...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
Standard penetration tests were carried out in normally consolidated fine sands at different locations. The following data were collected, with average unit weights assumed for the entire depth....
-
A standard penetration test was carried out in a normally consolidated sand at 7.62 m depth where the N 60 was determined to be 28. The unit weight of the sand is 17.29 kN/m 3 , and the grain-size...
-
Standard penetration tests were carried out in sands where the N 60 values at certain depths are reported as follows. The unit weight of the sand is 18.5 kN/m 3 . The water table is well below the...
-
Angela is entitled to a fixed annual payment of $15,000 from ABC Partnership without regard to the income of the partnership. Her share of the partnership income is 10%. After deducting the...
-
Rosie has $24,000 short-term capital loss, $10,500 28% gain and $9,000 0%/15%/20% gain. Which of the following statements is correct? a. Rosie has a $19,500 net capital gain b. Rosie has a $7,500...
-
Wyatt Company was formed on January 1, Year 1, when it acquired $50,000 cash from issuing common stock. Which of the following shows the impact of this transaction on Wyatt's accounting equation?...

Study smarter with the SolutionInn App


