(a) Compute e -a from Equation 8-44 for O 2 gas at standard conditions. (b) At what...
Question:
(a) Compute e-a from Equation 8-44 for O2 gas at standard conditions.
(b) At what temperature is e-a = 1 for O2?
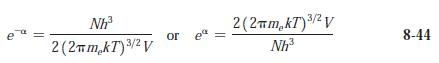
Transcribed Image Text:
e Nh³ 2 (2πm₂kT) ³/² V 3/2 or e 3/2 2(2πm₂kT) ³/² V Nh³ 8-44
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b a 0 N V hc h 2 MkT 3 NA V...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The following is a list of 11 measurements. 64,-88, -81, 43, -14, 61, -99, 80, -24, -45, 12 Suppose that these 11 measurements are respectively labeled r, X, ..., X (Thus, 64 is labeled x, 88 is...
-
At what temperature does the rms speed of O2 molecules equal 400. m/s?
-
A tank contains 26.0kg of O2 gas at a gauge pressure of 8.70 atm. If the oxygen is replaced by helium, how many kilograms of the latter will be needed to produce a gauge pressure of 7.00 atm?
-
At what points are the function. y = x tan x 2 x + 1
-
Sketch the normal distribution with a. = 3 and = 3. b. = 1 and = 3. c. = 3 and = 1.
-
1. Who are the key players in this industry? 2. If a price war will reduce margins, as the case suggests, why would any company embrace this strategy? 3. On what other strategy elements could the...
-
Describe how nonprofits intersect with your daily life.
-
Omega Corporation reports the following results for the current year: Gross profits on sales $120,000 Dividends from less-than-20%-owned domestic corporations . 40,000 Operating expenses 100,000...
-
Class 14 Pre-Class Quiz "If something cannot be objectively measured, it should be reported at its original purchase price in accordance with the
-
Pacific, Inc. is a technology consulting firm focused on website development and integration of Internet business applications. The president of the company expects to incur $ 775,000 of indirect...
-
Like 4 He, the most common form of neon, 20 Ne, is a rare gas and the 20 Ne atoms have zero spin and so are bosons. But unlike helium, neon does not become superfluid at low temperatures. Show that...
-
The molar mass of oxygen gas (O 2 ) is about 32 g/mol and that of hydrogen gas (H 2 ) about 2 g/mol. Compute (a) The rms speed of O 2 and (b) The rms speed of H 2 when the temperature is 0C.
-
Outline Speak digital marketing objectives for your organization.
-
Following are the average accounts receivable and net sales reported recently by two large beverage companies ( dollar amounts are stated in millions ) . Average Accounts Receivable Net Sales Molson...
-
Good summary of 200 words of ways in which I, as an HR professional demonstrate respectful and inclusive working, when clarifying problems and issues. For example, a description including one or more...
-
You are considering a project that requires a capital investment of $100 and will last for three years. Economical depreciation is straight line over 3 years (33.3% in each of the 3 years)) while tax...
-
If firms and workers have adaptive expectations, what impact will contractionary monetary policy have on inflation, unemployment, and the Phillips curve? If expectations are adaptive, how will the...
-
It is difficult to sue an auditor as a third party. Required Outline three key reasons why the third party might not succeed in litigation with the auditors.
-
Refer to the oscillator in Fig. 10.137. (a) Show that (b) Determine the oscillation frequency fo. (c) Obtain the relationship between R1 and R2 in order for oscillation to occur. R2 Ri L.
-
Why is it necessary to study the diffusion of molecules in biological systems?
-
a. Write a realistic problem for which this is the correct equation. Be sure that the answer your problem requests is consistent with the equation given. b. Finish the solution of the problem. 60 N =...
-
A 500 g steel block rotates on a steel table while attached to a 2.0-m-long massless rod. Compressed air fed through the rod is ejected from a nozzle on the back of the block, exerting a thrust force...
-
A 500 g steel block rotates on a steel table while attached to a 2.0-m-long massless rod. Compressed air fed through the rod is ejected from a nozzle on the back of the block, exerting a thrust force...
-
Problem 7-1 You are considering an option to purchase or rent a single residential property. You can rent it for $2,000 per month and the owner would be responsible for maintenance, property...
-
SHELL Evaluate the non-financial reporting practices and strategies adopted by the organisation, making reference to the reporting guidelines. - Make reference to what reporting frameworks the...
-
One symptom of the virus is mouth ulcers. As a result, you're now interested in investigating the oral condition of infected individuals. The data set contains data for a study of oral condition of...

Study smarter with the SolutionInn App


