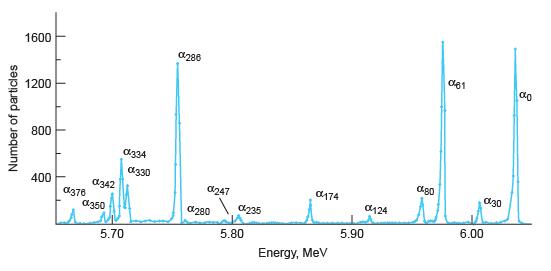
With the aid of Figures 11-19 and 11-20, list the energies of all of the possible g
Question:
With the aid of Figures 11-19 and 11-20, list the energies of all of the possible g rays that may be emitted by 223Ra following the a decay of 227Th.
Figures 11-19
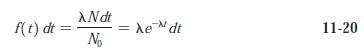
Transcribed Image Text:
Number of particles 1600 1200 800 400 I L T I 0376 0342 0350 0334 5.70 0330 0286 0247 280 0235 5.80 0174 Energy, MeV 5.90 0124 080 061 6.00 030 do
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Tabulated energies are in keV Higher energy levels in Figure 1119 wo...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
With the aid of the theorem in Sec. 17, show that when (a) (b) cd (ad lim T(z) = oo if c = 0;
-
With the aid of the result in Exercise 3, Sec. 38, evaluate the integral where m and n are integers and C is the unit circle |z| = 1, taken counterclockwise. mndz.
-
With the aid of a periodic table (not Figure 9.15), arrange the following in order of increasing electronegativity: a. Sr, Ca, Rb b. Ca, Ge, Ga c. Se, As, Sb
-
Factor the given expressions completely. 3a 3b
-
Determine the area under the standard normal curve that lies between a. 0.88 and 2.24. b. 2.5 and 2. c. 1.48 and 2.72. d. 5.1 and 1.
-
1. According to path-goal theory, why is Daniel an effective leader? 2. How does his leadership style affect the motivation of employees at The Copy Center? 3. How do characteristics of the tasks and...
-
Pyramid Construction Company is planning an investment of \($174,000\) for a bulldozer. The bulldozer is expected to operate for 1,400 hours per year for five years. Customers will be charged \($80\)...
-
Carpetland salespersons average $8,000 per week in sales. Steve Contois, the firms vice president, proposes a compensation plan with new selling incentives. Steve hopes that the results of a trial...
-
establishes on whose behalf a request for resources is made, whereas determines if the principle involved is allowed to have such access
-
The Jacksons are considering selling their current residence, buying a small home near Averys parents for $220,000 with a $100,000 30-year mortgage at 3.5%, and investing the net proceeds in their...
-
In Example 11-13 we saw that 233 Np could decay by emitting an a particle. Show that decay by emission of a nucleon of either type is forbidden for this nuclide.
-
Compute the maximum energy of the - particle emitted in the decay of 72 Zn.
-
Discuss and give an example of one-to-one, one-to-many, and many-to-many record associations.
-
what is contract services cost of debit, asset beta, levered beta, cost of equity, and WACC ? Explain
-
A 10 mm thick copper plate is cut using wire EDM. If the kerf width is 1 mm, the specific heat is 1550 J/g, and the density is 8970 kg/m3, How fast can the wire move in mm/swhile maintaining the...
-
why can FinTech have important risk management implications for financial institutions?
-
A company expects a 2 0 % return on equity invested entirely in new projects. The company's management plans to retain 3 0 % of its net profits. Earnings per share for the coming year are 3 and the...
-
Establish a branding model, which establishes stages and functions, and which promotes a development of tactics regarding branding of a brand.
-
Using mesh analysis, obtain I o in the circuit shown in Fig. 10.83. Io 2402 A ( ) 10,790" V -/4 1S2 112
-
Should we separate the debt and equity features of convertible debt? Team 1: Pro separation: Present arguments in favor of separating the debt and equity features of convertible debt. Team 2: Against...
-
A positive pion at rest decays into a positive muon and a neutrino. (a) Approximately how much energy is released in the decay? (Assume the neutrino has zero rest mass. Use the muon and pion masses...
-
Model a hydrogen atom as an electron in a cubical box with side length L. Set the value of L so that the volume of the box equals the volume of a sphere of radius a = 5.29 x 10 -11 m,, the Bohr...
-
(a) Write out the ground-state electron configuration (1s 2 , 2s 2 ,) for the carbon atom. (b) What element of next-larger Z has chemical properties similar to those of carbon? Give the ground-state...
-
II. Roger's Manufacturing Company is selling its classic Rolls-Royce Phantom in mint condition for $200,000 at a public auction. The car was originally bought for $60,000 and it has been always kept...
-
The RLX Company just paid a dividend of $3.05 per share on its stock. The dividends are expected to grow at a constant rate of 5.5 percent per year, indefinitely. Assume investors require a return of...
-
Describe the topic you have chosen to study and explain what kinds of data you will collect to present to your government representative. Describe two specific recommendations you would make to...

Study smarter with the SolutionInn App


