Work out the quadratic Zeeman effect for the ground-state hydrogen atom [(x|0) = (1/a 0 3 )
Question:
Work out the quadratic Zeeman effect for the ground-state hydrogen atom [(x|0) = (1/√πa03) e-r/a0] due to the usually neglected e2A2/2mec2- -term in the Hamiltonian taken to first order. Write the energy shift as![]()
and obtain an expression for diamagnetic susceptibility, X. The following definite integral may be useful: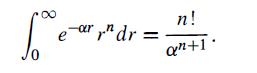
Transcribed Image Text:
A=-1/XB²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The quadratic Zeeman effect refers to the splitting of atomic energy levels in the presence of a mag...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The hydrogen atom is composed of one proton in the nucleus and one electron, which moves about the nucleus. In the quantum theory of atomic structure, it is assumed that the electron does not move in...
-
Consider Devine Fashion from S8-6. Assume that the fixed expenses assigned to each department include only direct fixed costs of the department (rather than unavoidable fixed costs as given in S8-6):...
-
Explain the technique known as destructive update.
-
Your best friends age. In Exercises 1524, determine whether the data described are qualitative or quantitative.
-
The FASB states in SFAC 1: Information about enterprise earnings based on accrual accounting generally provides a better indicator of an enterprise's present and continuing ability to generate...
-
Outdoor Inn, a camping equipment manufacturer in southern Utah, is developing a production schedule for a popular type of tent, the Double Inn. Orders have been received for 180 of these to be...
-
Please show work. Question 3 of 13 5 Points You are choosing between three 1-year investments: one in Vietnam, one in Thailand, and one in Japan. The investment in Vietnam yields a nominal rate of...
-
Suppose the electron had a very small intrinsic electric dipole moment analogous to the spin-magnetic moment (that is, el proportional to ). Treating the hypothetical - el . E interaction as a small...
-
Work out the Stark effect to lowest nonvanishing order for the n = 3 level of the hydrogen atom. Ignoring the spin-orbit force and relativistic correction (Lamb shift), obtain not only the energy...
-
On October 1, Chile Corporations stockholders equity is as follows. Common stock, $5 par value ......... $200,000 Paid-in capital in excess of par value ...... 25,000 Retained earnings .................
-
After reading the Legacy of Court TV (Box 6.6) discuss the positive and negative effects of televising criminal trials. How does this compare to crime dramas that are focused on the courtroom....
-
When dealing with other workers, provide an example of how to respect their differences in culture.
-
You are a lawyer or paralegal in a firm that represents Veterans in front of the Board of Veterans Appeals (BVA). You just got a new client, Ed, who fought in the Vietnam War. At the time, he applied...
-
When deciding the best strategy to add extra time on a project, we should consider an approach that considers the cost of overtime the amount of time to be reduced lighting requirements work morale...
-
Patterson Electronics supplies microcomputer circuitry to a company that incorporates microprocessors into refrigerators and other home appliances. One of the components has an annual demand of 250...
-
Bermuda residents will be able to transfer their money to almost anyone in the world via text messaging, thanks to a new service being offered by Bermuda Financial Network Ltd. If Canadian residents...
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
What is the formal concentration (expressed as mol/L = M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?
-
How many grams of boric acid, B(OH) 3 (FM 61.83), should be used to make 2.00 L of 0.050 0 M solution? What kind of flask is used to prepare this solution?
-
Describe how you would prepare approximately 2 L of 0.050 0 m boric acid, B(OH) 3 .
-
b 2.5 points Required information Use the following information for the Exercises below. (Algo) [The following information applies to the questions displayed below) Manuel Company predicts it will...
-
Add JUnit testing to "word frequency" class. include tests to verify that the class correctly counts the occurrences of words in a .txt file, handles edge cases such as empty input or non-existent...
-
. Authors: Sabina, Daniel, Eric Topics: polymorphism, exceptions, file l/O Problem Description Please make sure to read all parts of this document carefully. You and your friends are big fans of...

Study smarter with the SolutionInn App


