Use the data given in Table 9.2 to find the dissociation energies D e and D 0
Question:
Use the data given in Table 9.2 to find the dissociation energies D̃e and D̃0 (in units of cm−1) for 2H35Cl.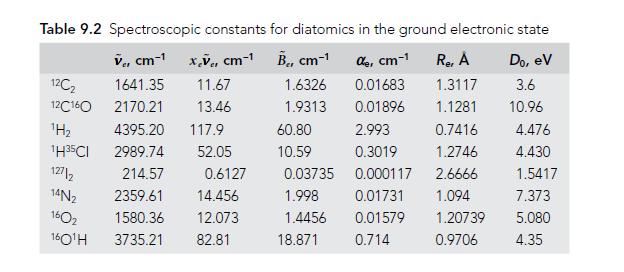
Transcribed Image Text:
Table 9.2 Spectroscopic constants for diatomics in the ground electronic state V, cm-¹x,V, cm-¹ B₁, cm-¹ de, cm-1 1.6326 0.01683 1.9313 0.01896 12C₂ 12C160 ¹H₂ 1H35CI 127/2 14N₂ 160₂ 160¹H 1641.35 11.67 2170.21 13.46 4395.20 117.9 2989.74 52.05 214.57 2359.61 14.456 1580.36 12.073 3735.21 82.81 0.6127 60.80 10.59 0.03735 1.998 1.4456 18.871 Re, A Do, ev 1.3117 3.6 1.1281 10.96 2.993 0.7416 0.3019 1.2746 0.000117 2.6666 0.01731 1.094 0.01579 0.714 1.20739 0.9706 4.476 4.430 1.5417 7.373 5.080 4.35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
To find the dissociation energies De and D0 in units of cm for 2H35Cl we can ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the data given in the previous exercise to complete the following: a. Estimate the mean difference in motion between dominant and nondominant arms for position players using a 95% confidence...
-
In Cylindrical coordinates: For p < 2m, Er = 2 and E = 6p + 5g + 9, V/m. For p < 2m, Er2 =3. Determine E,
-
Use the data given in the following table and consider the following model: ln Savings i = β 1 + β 2 ln Income i + β 3 ln D i + u i where ln stands for natural log...
-
Modify BST to add a method rangeSearch () that takes two keys as arguments and returns an iterable over all keys that are between the two given keys. The running time should be proportional to the...
-
Find the water pressure at ground level to supply water to the third floor of a building 8.00 m high with a pressure of 325 kPa at the third-floor level.
-
How many stationary policies are there in the problem of Exercise 5?
-
\(\sqrt{144}\) Identify which of the following are rational numbers.
-
Presented below are two independent situations. (a) On January 6, Brumbaugh Co. sells merchandise on account to Pryor Inc. for $7,000, terms 2/10, n/30. On January 16, Pryor Inc. pays the amount due....
-
Case - To Sell or Not to Sell The CFO of X, a brokerage firm and SEC registrant (the "Company"), tells you that the Company is contemplating a sale of one of its reporting units, G, a wholly owned...
-
The vibrational frequency of 1 H 79 Br is v e =2649.7cm 1 and the anharmonicity is x e v e =452.cm 1 . Find the frequencies of the fundamental, first overtone and second overtone for 1 H 79 Br, 2 H...
-
Show that the dipole moment of a collection of charges is independent of origin, provided that the net charge is zero. Also, show that the quadrupole moment is independent of origin if the dipole...
-
During June, Lewis Company accumulated 1,200 hours of direct labor costs on Job 30 and 1,300 hours on Job 32. The total direct labor was incurred at a rate of $16 per direct labor hour for Job 30 and...
-
Who are some of the outside groups that may be interested in a company's financial data and what are their particular interests?
-
With regard to the taxation of the sale of software: A. Is there a clear distinction between canned and custom soft ware? Is there any valid reason for drawing the distinction be tween canned and...
-
What is the true object test? In what connection has this test been used? How have the courts operationalized this test? Apart from the cases discussed in the text, locate one or more cases in your...
-
Puff Company began the year with a retained earnings balance of \(\$ 50,000\), reported net income for the year of \(\$ 45,000\), and reported ending retained earnings of \(\$ 60,000\). How much...
-
What is the rationale, if any, for states to generally exclude services from their sales tax base?
-
Assume that a central banks nominal seigniorage revenue equals the change in the money supply, denoted AM. Real seigniorage revenue is AM/P. Assume the inflation rate equals the growth rate of the...
-
Write each fraction as a percent. 7 50
-
Identify the reagents necessary to achieve each of the following transformations: Br Br - " Br Br "Br
-
Determine the configuration for every chirality center in each of the following compounds. a. b. c. - - - - - - CH- - CH- II
-
For each of the following reactions predict the sign of G. If a prediction is not possible because the sign of G will be temperature dependent, describe how G will be affected by raising the...
-
A partir del 31 de diciembre de 2013, Zintel Corporation proponeemitir acciones adicionales de sus acciones comunes a cambio detodos los activos y pasivos de Smith Corporation y PlatzCorporation, l 1...
-
Periodic Inventory by Three Methods The units of an itemavailable for sale during the year were as follows: Jan. 1Inventory 5 units at $41 Feb. 17 Purchase 10 units at $43 Jul. 21Purchase 5 units a 2...
-
based on the period of 1926 through 2005 _________ have tendedto out perform other securities over the long terma) large company stocksb) long term government bondsc) small company stocksd) U.S t 2...

Study smarter with the SolutionInn App


