Acetals can serve as protecting groups for 1,2-diols, as well as for aldehydes and ketones. When the
Question:
Acetals can serve as protecting groups for 1,2-diols, as well as for aldehydes and ketones. When the acetal is formed from acetone plus the diol, the acetal is called an acetonide. Show the acetonides formed from these diols with acetone under acid catalysis.
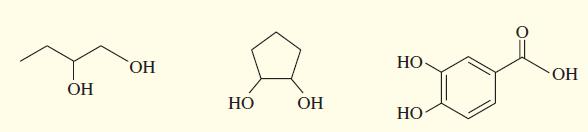
Transcribed Image Text:
HO, НО- OH OH НО OH НО
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a b c Mechanism H o...View the full answer

Answered By

Nakul Naskar
I am a organic chemist. I have experience of teaching organic chemistry to bring out Concepts about this subject which helps a student to enhance his/her knowledge and make a good impact on his educational life. Besides I can also teach Inorganic and physical chemistry.
I can monitor student performance or assist students in academic environments, such as classrooms, laboratories.
By Providing feedback to students using positive reinforcement techniques to encourage, motivate, or build confidence in students.
Prepare lesson plans or learning modules for tutoring sessions according to students' needs and goals.
During my teaching life I Maintain records of students' assessment results, progress, feedback, or school performance, ensuring confidentiality of all records.
I can clear your all doubts on chemistry by providing easy explanation.
Thank you
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Acetal formation is a characteristic reaction of aldehydes and ketones, but not of carboxylic acids. Show how you could advantageously use a cyclic acetal protecting group in the following synthesis:
-
Acetal formation is a characteristic reaction of aldehydes and ketones, but not of carboxylic acids. Show how you could advantageously use a cyclic acetal protecting group in the following synthesis:...
-
Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism: H* catalyst 2...
-
The voltage held by a voltage regulator follows a normal random variable with a mean that equals 200 volts and a standard deviation that equals 5 volts. A regulator meets the specifications if the...
-
Use electron dot structures to show why tetramethylammonium hydroxide, (CH3)4N+OH-, is an ionic compound. That is, show why hydroxide is not covalently bound to the rest of the molecule.
-
Derive appropriate mass balance equation and solutions equivalent to Eqs. (20-22) to (2024) but for the case where \(u_{S}\left(T_{\text {hot }} ight)>u_{S}\left(T_{\text {cold }} ight)>u_{\text {th....
-
Krispy Kreme Doughnuts, Inc. has franchises operating outside the U.S. and invests in derivatives to hedge its foreign currency risk. Although it tries to hedge its various positions, on December 16,...
-
Alternative allocation bases for a professional services firm. The Walliston Group (WG) provides tax advice to multinational firms. WG charges clients for (a) Direct professional time (at an hourly...
-
Do you have any exposure to sales accounting in your current position or through a role that you held in the past? Provide the company name, a few sentences about what they do, a link to their...
-
Parentis, a public listed company, acquired 600 million equity shares in Offspring on 1 April 2006. The purchase consideration was made up of: - A share exchange of one share in Parentis for two...
-
Rank the following carbonyl compounds in order of increasing equilibrium constant for hydration: CH,COCH,CI CICH,CHO CH,0 CH3COCH3 CH3CHO
-
Sketch the expected proton NMR spectrum of 3,3-dimethylbutanal.
-
The following set of scores was obtained from a quiz: 4, 5, 8, 9, 11, 13, 15, 18, 18, 18, 20. The teacher computes the usual descriptive measures of central tendency and spread for these data and...
-
Is impression management negative or positive? Why?
-
Why do economists sometimes conduct analysis under assumptions of a closed economy? What do you understand by the term an open economy?
-
Discuss the types of reinforcements that are available to managers for changing an employees behavior.
-
Explain how goals can be determined under the Goal-Setting Theory.
-
The economist John Maynard Keynes wrote: Lenin is said to have declared that the best way to destroy the capitalist system was to debauch the currency. By a continuing process of inflation,...
-
Would a tax year ending December 31 be appropriate for a Ski lodge? Why or why not?
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
Arrange the following compounds in order of increasing reactivity toward HNO3 in H2SO4. (The references to equations will assist you with nomenclature.) (a) Chlorobenzene, benzene, nitrobenzene (b)...
-
Rank the following compounds in order of increasing reactivity in bromination. In each case, indicate whether the principal monobromination products will be the ortho and para isomers or the meta...
-
Rank the following compounds in order of increasing reactivity in bromination. In each case, indicate whether the principal monobromination products will be the ortho and para isomers or the meta...
-
10.8 Staht Company was incorporated as a new business in January. The company is authorized to issue 600,000 shares of \( \$ 2 \) par value common stock and 80,000 shares of \( 6 \%, \$ 20 \) par...
-
On April 1, 2023, ET Inc. has available for issue $216,000 bonds due in four years. Interest at the rate of 4.5% is to be paid quarterly. Calculate the issue price if the market interest rate is: (Do...
-
The following events apply to Paradise Vacations's first year of operations: 1. Acquired $30,000 cash from the issue of common stock on January 1, Year 1. 2. Purchased $1,300 of supplies on account....

Study smarter with the SolutionInn App


