Alkenes undergo reaction with peroxycarboxylic acids to give epoxides. For example, cis-but-2-ene gives 2,3-epoxybutane: H3C
Question:
Alkenes undergo reaction with peroxycarboxylic acids to give epoxides. For example, cis-but-2-ene gives 2,3-epoxybutane:
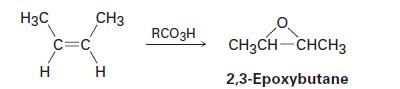
Transcribed Image Text:
H3C Н Н C=C CH3 Н RCO3H CH3CH-CHCH3 2,3-Epoxybutane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Thats correct Alkenes can undergo a reaction with peroxycarboxylic acids to form epoxides a t...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structures of all epoxides that could in principle be formed when each of the following alkenes reacts with meta-chloroperoxybenzorc acid (mCPBA). Which epoxide should predominate in each...
-
Alkenes slowly undergo a reaction in air called autoxidation in which allylic hydroperoxides are formed. OOH Cyclohexene Oxygen 3-Hydroperoxycyclohexene 0:0)
-
Give the structures of the two stereoisomeric alkenes with the molecular formula C6H12 that react with HI to give the same single product and undergo catalytic hydrogenation to give hexane.
-
Perform the indicated operations and then simplify. Assume that all variables represent nonzero real numbers. 20yx3 + 15y4x + 25yx4 10yx
-
You are a bidder in an independent private values auction, and you value the object at $ 4,000. Each bidder perceives that valuations are uniformly distributed between $ 1,500 and $ 9,000. Determine...
-
If you were to guess, what percentage of social entrepreneurs engage in formal assessment of their ideas as compared with those who do so informally? Explain your reasoning. What are the advantages...
-
On January 1, 2020, Josie Inc. purchased for cash ten, \(\$ 1,000,4 \%\) bonds of Star Corp. at par. The bond interest is paid annually on January 1 of each year, and the bonds mature on January 1,...
-
Companion Computer Company has been purchasing carrying cases for its portable computers at a delivered cost of $68 per unit. The company, which is currently operating below full capacity, charges...
-
J. Lo??s Clothiers has forecast credit sales for the fourthquarter of the year:September (actual)$62,000Fourth QuarterOctober$52,000November47,000December72,000Ex 2 answers
-
We said in Section 4.9 that an allylic carbocation is stabilized by resonance. Draw resonance structures to account for the similar stabilization of a benzylic carbocation. + CH A benzylic carbocation
-
Draw the two cistrans stereoisomers of 1,2 dimethyl cyclopentane, assign R,S configurations to the chirality centers, and indicate whether the stereoisomers are chiral or meso.
-
Valley Fruit Products Company has contracted with apple growers in Ohio, Pennsylvania, and New York to purchase apples that the company then ships to its plants in Indiana and Georgia, where they are...
-
Peter Easton, the CEO of EWHM , did not sleep all night. He knew he was about to lose his lucrative position. And that was so unfair. He had worked so hard the entire year - and delivered. But the...
-
John Mayor is an analyst reviewing his valuation of PWS Insurance (PWS).He has the following information and assumptions regarding PWS. The current market price is R67.53. Mayor's estimate of PWS's...
-
Determine if each function is one-to-one or not. f(x)=7x-5 Select an answer 7x 5 g(x) x+6 Select an answer h(x) = 7x25 Select an answer
-
Apple is planning to incorporate new software for the price of $ 5 0 0 0 0 0 today in order to produce a new line of the iPod. The new iPod will be ready for sale in 1 year. If the new software will...
-
Thulasizwe's shares are selling for R72 today and have paid a dividend of R1.50 per share for the year 2020. The required rate of return on equity is 10.50%. What is the constant dividend growth rate...
-
What are the main differences between currency swaps, options, and futures?
-
A heat engine has a heat input of 3 Ã 104 Btu/h and a thermal efficiency of 40 percent. Calculate the power it will produce, in hp. Source 3 x 10 Btu/h 40% HE Sink
-
Consider the structure of N,N-dimethylformamide (DMF): We might expect the two methyl groups to be equivalent; however, both the proton and carbon NMR spectra of DMF show two separate signals for the...
-
Consider the structure of phenol: The chemical shift of the hydroxyl proton is found to be sensitive to the concentration of phenol. In a concentrated solution, the hydroxyl proton produces a signal...
-
Consider the two methyl groups shown in the following compound. Explain why the methyl group on the right side appears at lower chemical shift. CH3 0.8 ppm 1.0 ppm H;C.
-
The cost of materials for beginning work-in-process is $10,800. Materials costs added during the period were $131,200. The equivalent units of production for materials is 53,000 units. Using the...
-
sa senior debt-holder, which of these would be a high risk indicator and would require further investigation when monitoring financial statements? OA reduction in the total long-term liabilities. O...
-
Tax liability\ $ \ 105,630,000 \ Determine the firm's net income.\ Note: Enter your answer in dollars not in millions.\ Hint\ Net income\ 397,370,000 \ Determine the firm's average tax rate.\ Average...

Study smarter with the SolutionInn App


