An unknown compound gives the following mass, IR, and NMR spectra. Propose a structure, and show how
Question:
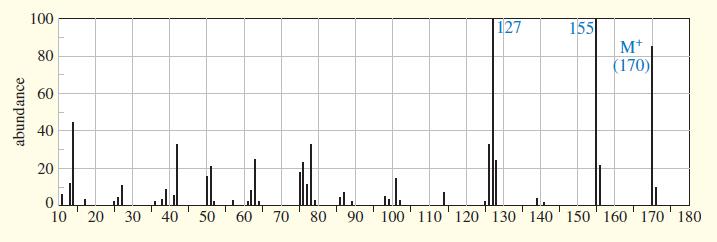
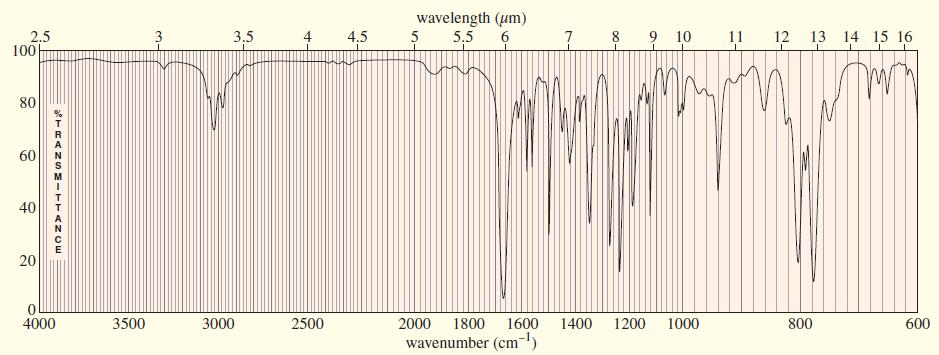
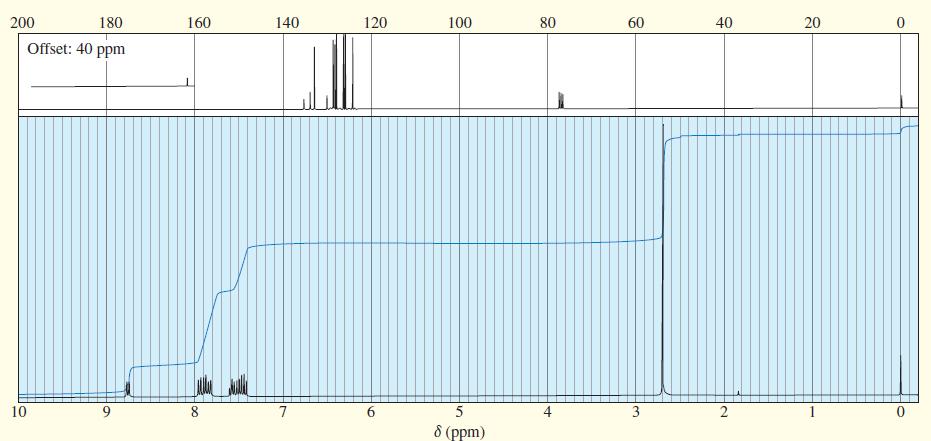
An unknown compound gives the following mass, IR, and NMR spectra. Propose a structure, and show how it is consistent with the spectra. Show the fragmentations that give the prominent peaks at m>z 127 and 155 in the mass spectrum.
Transcribed Image Text:
100 155 M+ (170) 127 80 60 40 20 10 20 40 30 50 60 70 80 ' 90 '100 ' 110 120' 130' 140 150' 160' 170 180 abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
The mass spectrum shows a molecular ion at 170 This serves as the molecular weight for the compound While as is typical the fragmentation is complex t...View the full answer

Answered By

NEHA SINGH
I have done my bacheoler' from DEEN DAYAL UAPADHAYAY GORAKHPUR UNIVERSITY. I am pursuing my master's from IIT BHU. I am former online tutor on CHEGG INDIA. I really enjoying to teach and help students .it is so satisfactory
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The elemental analysis of an unknown compound gives the following results: 74.99 % C, 5.034 % H, 19.98 % O. What is the empirical formula of the unknown compound
-
An unknown compound gives the NMR, IR, and mass spectra shown next. Propose a structure, and show how it is consistent with the observed absorptions. Show fragmentations that account for the...
-
An unknown compound gives a mass spectrum with a weak molecular ion at m/z 113 and a prominent ion at m z 68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is...
-
How are financial statements adjusted for exchange rates?
-
Write the Kb reactions for pyridine and for sodium 2 - mercap - toethanol. ON HOCH,CH,S: Na Pyridine Sodium 2-mercaptoethanol
-
We are doing RO experiments with a completely mixed laboratory unit. Data: \(\mathrm{K}_{\text {solv }}^{\prime} / \mathrm{t}_{\mathrm{ms}}=1.387 \mathrm{~g} /\left(\mathrm{atm} \mathrm{m}^{2}...
-
5. What can you do to protect your privacy online? Draw up a concrete list of steps that you might reasonably consider. Are there some actions that you would not be willing to take, either because...
-
On January 1, 2017, Geffrey Corporation had the following stockholders equity accounts. Common Stock ($20 par value, 60,000 shares issued and outstanding).. $1,200,000 Paid-in Capital in Excess of...
-
MC Qu. 101 The statement of cash flows helps... < > h newconnect.mheducation.com C + Exam 3B (40) i Saved Help Save & Exit Submit 39 MC Qu. 176 Northington, Inc. is preparing.. Northington, ...
-
Tomato carries on a business of tomato faming. Its financial year ends on the last day of February. In January 2018 Tomato sold, and delivered, tomatoes to a tomato-sauce producer for R 25 000. The...
-
A student found an old bottle labeled thymol on the stockroom shelf. After noticing a pleasant odor, she obtained the following mass, IR, and NMR spectra. The NMR peak at 4.8 disappears on shaking...
-
Propose a mechanism for each cyanohydrin synthesis just shown.
-
(a) Discuss the use of the following as aids to each of planning and control: (i) rolling budgets; (ii) flexible budgets; (iii) planning and operational variances. (b) Discuss the extent to which the...
-
What is the purpose of a drilling machine ? Explain its working principle.
-
How the size of milling machine is specified?
-
Describe the construction and working of the following : (a) General purpose milling machine. (b) Vertical milling machine.
-
List the various types of milling machines.
-
What are the main functions of a milling machine?
-
Which of the following is excluded from gross income on an individual's 2016 tax return? a. 2016 settlement from an employment discrimination lawsuit b. Seller of the Year award, in the form of a...
-
Match each of the key terms with the definition that best fits it. _______________ A record of the sequence of data entries and the date of those entries. Here are the key terms from the chapter. The...
-
A small amend of a by4roduct, gdibromobenzene, is also formed in the brorlination of benzene shown in Eq. 16.2. Write a curved-arrow mechanism for formation of this compound. Eq. 16.2 HBr FeBra or Fe...
-
A compound called p-toluenesulfonic acid is formed when toluene is sulfonated at the para position. Draw the structure of this compound, and give the curved-arrow mechanism for its formation.
-
What electrophilic substitution product is formed when 2-methylpropene is added to a large excess of benzene containing HF and the Lewis-acid BF3? By what mechanism is it formed?
-
Bridgeport Company exchanges old delivery equipment for newdelivery equipment. The book value of the old delivery equipment is$32,000 (cost $62,300 less accumulated depreciation $30,300). Itsfair v 2...
-
Job Number 544 $ 15,600 545 A company reports the following information on April 30, the end of its first month of operations. Manufacturing Costs Job Status at April 30 Completed and sold during...
-
Under a modified wage plan, Jim Phillips works an eight-hour day and earns $.50 for each finished unit he produces in excess of 200 units. However, he is guaranteed $12.50 per hour as a minimum wage....

Study smarter with the SolutionInn App


