Convert the following perspective formulas to Fischer projections. (a) (b) (c) (d) H OH .C. CH; CH,OH
Question:
Convert the following perspective formulas to Fischer projections.
(a)
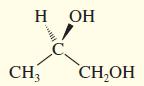
(b)
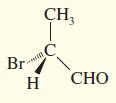
(c)
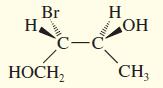
(d)
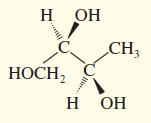
Transcribed Image Text:
H OH .C. CH; CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
1 2 3...View the full answer

Answered By

Vartika ..
Having secured 10 CGPA during school level education, and being one of the toppers in my school as well as being college topper with distinction, I have always conceptualised chemistry not just as a subject, but also as a mystery to explore more about.
I have been teaching since college days and have helped students clear their doubts as well as exams.
The biggest compliment i received was when one of my students said that I answer even those queries, which even their teachers couldn't answer.
I welcome you all to come with me and explore the mysterious world of chemistry ;)
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw enantiomers for each of the following compounds using: a. perspective formulas b. Fischer projections CH CH 1. CHjCHCH2H 2. CICH2CH2CHCH2CH3 3. CHjCHCHCHj
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you have drawn. a) CH OH
-
In Exercises use a graphing utility to graph the curve represented by the parametric equations. Indicate the direction of the curve. Identify any points at which the curve is not smooth. Curtate...
-
Why does buffer capacity increase as the concentration of buffer increases?
-
Let \(\sim\) be an equivalence relation on \(\mathbb{Z}\) with the property that for all \(m \in \mathbb{Z}\) we have \(m \sim m+5\) and also \(m \sim m+8\). Prove that \(m \sim n\) for all \(m, n...
-
5. YOU BE THE JUDGE WRITING PROBLEM Dominion Bank owned a large office building in Washington, D.C. Because it planned to sell the building, the bank stopped leasing new space and five of the 13...
-
Here is financial information for Spangles Inc. Instructions Prepare a schedule showing a horizontal analysis for 2014, using 2013 as the baseyear. December 31, 2014 106,000 400,000 99,000 122,000...
-
The company, North S.L., has the following information in the Balance Sheet and Profit and Loss Account for 2016, before calculating its Corporate Tax: North S.L. is thinking of making an...
-
Read Using Cost-Volume-Profit Analysis in Decision Making. The authors mention that cost-volume-profit (CVP) analysis is only useful in certain circumstances and when certain assumptions are valid....
-
A chiral sample gives a rotation that is close to 180. How can one tell whether this rotation is +180 or -180?
-
Give the stereochemical relationships between each pair of structures. Examples are same compound, structural isomers, enantiomers, diastereomers. Which pairs could you (theoretically) separate by...
-
How should a market entry strategy be determined? What factors should be considered?
-
What is the Hague Convention on the Civil Aspects of International Child Abduction?
-
What is covenant marriage? What do supporters hope to accomplish?
-
How do guidelines treat the income of a child?
-
Explain the no-fault ground of living separate and apart.
-
Do you think the emergency jurisdiction provision of the UCCJEA might encourage parents to flee with children in the absence of a genuine emergency situation in order to try and gain an advantage in...
-
"Wolfram Putz et.al" read this case and answers the following: What is the Innovation/Service that Placed, Explain the Innovation and discuss more about the firm/person beyond what you read in this...
-
In Exercises 105108, evaluate each expression without using a calculator. log(ln e)
-
Predict the relative intensities of the three peaks in the mass spectrum of dichloromethane at m/z = 84, 86, and 88.
-
From the molecular masses and the relative intensities of their M and M + 1 peaks, suggest molecular formulas for the following compounds. M (m/z = 82; 37%), M + 1 (2.5%); contains C and H.
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
Question 13 The debt ratio is used: D. To determine how much debt a firm should pay off. To measure the ratio of equity to expenses. To assess the risk associated with a company's use of liabilities....
-
Question 1 Trial Balance for the financial year (FY) ended 31st March 2022 of M/s Deepakshi shows following details: Particulars Debit (`) Credit (`) Purchase & Sales 10,00,000 12,00,000 Debtors &...
-
Course Content / MA - Chapter 6 MA Chapter 6 Homework Question 2 Not changed since last attempt < Previous Marked out of 3.30 Save Answers MA Chapter 6 Homework P Flag question eBook Total Cost...

Study smarter with the SolutionInn App


