For each molecule shown below, 1. indicate the most acidic hydrogens. 2. draw the important resonance contributors
Question:
For each molecule shown below,
1. indicate the most acidic hydrogens.
2. draw the important resonance contributors of the anion that results from removal of the most acidic hydrogen.
a.
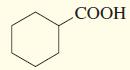
b.
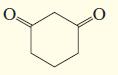
c.
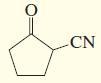
d.
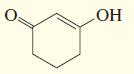
e.
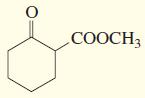
f.
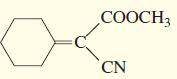
g.
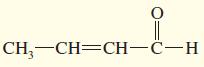
h.
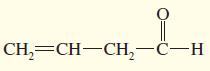
Transcribed Image Text:
СООН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
A The given compound is cyclohexanoic acidThe most acidic hydrogen in that compound is carboxylic group of hydrogen due to the hydrogen is attached to ...View the full answer

Answered By

KATTUBOINA KOTA SATYA SAIKUMAR
EDUCATION;
I qualified CSIR-NET EXAM in december 2019.i got 49th rank in LS category,and also i cleared APSET exam which is conduct by ANDHRA UNIVERSITY in 2018.
M.SC (chemistry) from ANDHRA UNIVERSITY 2016-2018.
B.SC (MPC) from ADIKAVINANNAYYA UNIVERSITY 2013-2016.
Now i am working as chemistry lecturer in s.m.v.m.polytechnic college .
i can teach chemistry in iit-jee level and also i solve chemistry problems as simple as possible and give tricks to students for solve problems.i also help to students for enhancing their chemistry subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the important resonance contributors for the following cations, anions, and radicals. (a) (b) (c) (d) (e) (f) (g) (h) CH CH 2 - CH2 OF OCH3
-
Draw the important resonance contributors for both resonance-stabilized cations (in brackets) in the mechanism for acid-catalyzed hydrolysis of an amide.
-
Draw the important resonance forms to show the delocalization of charges in the following ions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH-C CH H-C-CH CHCH CH2 NH CHCH- CH CH CH-CH 3 CH3 CH CH CH CH...
-
Use implicit differentiation to find dy/dx. 6x 3 + 7y 3 = 13xy
-
A solution containing 3.47 mM X (analyte) and 1.72 mM S (standard) gave peak areas of 3 473 and 10 222, respectively, in a chromatographic analysis. Then 1.00 mL of 8.47 mM S was added to 5.00 mL of...
-
Accounts Payable is an example of an asset account. True/False
-
1.15 Euro Virtual's EPS Sensitivity to Exchange Rates (B). Assume a major weather crisis hits Switzerland, reducing its agricultural and food industries and sub- sequently leading to a macroeconomic...
-
The cash account for Collegiate Sports Co. on November 1, 2014, indicated a balance of $81,145. During November, the total cash deposited was $293,150, and checks written totaled $307,360. The bank...
-
MC Qu. 88 Which one of the following is representative... H newconnectmheducation'com Exam 3B (40) G Help Save 5 Exit Submit 3 8 MC On. 38 Which one of the following ls representative... Which one...
-
Suppose a community has 1000 families living in it. What is the probability that between one and five families are hypertensive? Hypertension People are classified as hypertensive if their systolic...
-
Show how you would use Robinson annulations to synthesize the following compounds. Work backward, remembering that the cyclohexenone is the new ring and that the double bond of the cyclohexenone is...
-
Show how you would accomplish the following multistep conversions. You may use any additional reagents you need. a. b. c. d. dimethyl adipate and allyl bromide
-
A column in the Wall Street Journal argues that many firms have shifted away from traditional advertising toward providing content: Content, these days, is the catchall term for all the media that we...
-
Why might herd mentality lead to asset prices rising faster than the fundamental value of the assets?
-
What are the fundamental assumptions and approaches of classical and neo-classical economics?
-
Why can mental accounting lead to irrational decision-making?
-
A firms strategy should describe how it intends to create and sustain value for what entities?
-
The fiscal compact would be fine if all countries in the euro area were at the same stage in the economic cycle and it was designed to act as a deterrent for profligacy in the future. As a means of...
-
Michael and Kathy have one dependent, Dustin, who is in his third year of college. Michael is taking classes in the evening toward an MBA. What credits can Michael and Kathy claim as to these...
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Write a curved-arrow mechanism for the hydroxide-catalyzed hydration of acetaldehyde. (Eq. 19.15) pH 9-10 acetone C N acetone cyanohydrin (77-78% yield)
-
Write a curved-arrow mechanism for The methoxide-catalyzed addition of methanol to benzaldehyde.
-
Which carbonyl compound should form the greater proportion of cyanohydrin at equilibrium? Draw the structure of the cyanohydrin, and explain your reasoning. CH-O or CH,CH,CH propanal benzaldehyde
-
Can you please Write a Java code: Library has a name (String), foundation year (int), number of books (int) and book array variables where all of them are private. ? Book has a name (String), publ...
-
ANSWERS only PLEASE The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods: Current assets as of March 31: Cash $ 8,700 Accounts receivable...
-
Two products, QI and VH, emerge from a joint process. Product QI has been allocated $18,300 of the total joint costs of $39,000. A total of 2,500 units of product QI are produced from the joint...

Study smarter with the SolutionInn App


