How would you prepare the following alkyl halides from the appropriate alcohols? (a) CI CH3CCH3 CH3 CH3
Question:
How would you prepare the following alkyl halides from the appropriate alcohols?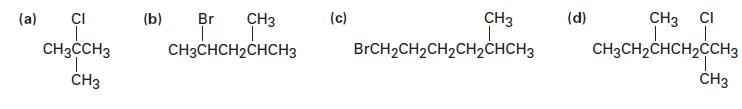
Transcribed Image Text:
(a) CI CH3CCH3 CH3 CH3 ī CH3CHCH₂CHCH3 (b) Br (c) CH3 T BrCH₂CH₂CH₂CH₂CHCH3 (d) CH3 CI I I CH3CH₂CHCH₂CCH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To prepare the given alkyl halides from the appropriate alcohols we need to perform a substitution r...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you prepare the following alkyl halides from the correspondingalcohols? CH (a) CI (b) Br CH CHH2CH CH (c) CI H (d) BrCH2CH2CH2CHCH CHCH2H2cH3 CH
-
Which of the following alkyl halides form a substitution product in an SN1 reaction that is different from the substitution product formed in an SN2 reaction? a. b. c. d. e. f. CH Br CH CHCHCHCH CHa...
-
How many alkyl halides can be obtained from monochlorination of the following alkanes? Neglect stereoisomers. (a) CH3CH2CH2CH2CH3 (b) (c) (d) (e) (f) (g) (h) (i) CH3 H3 CH CHCH2CH2CHCH3 , CH3 CH3...
-
For Problem 511, complete the design of a pair of helical gears to operate under the stated conditions. Specify the geometry of the gears and the material and its heat treatment. Assume that the...
-
A firm produces output according to the production function Q = F (K, L) = 4K + 8L. a. How much output is produced when K = 2 and L = 3? b. If the wage rate is $ 60 per hour and the rental rate on...
-
Calculate the molar solubility of silver iodide, AgI, in 2.2 M NH 3 .
-
Expected value Gasoline sales by a service station average $1,200 a day on clear days, but only $500 on rainy days. The weather forecast for tomorrow is for a 70 percent chance of thundershowers....
-
Big Bend Photo Shop has asked you to determine whether the companys ability to pay current liabilities and total liabilities improved or deteriorated during 2016. To answer this question, you gather...
-
Lec14 ( X G what ar X Lewis S X PF5 Lev X Lewis [ X G ammor X Edmon X Assign X G inverse X Desmo X Homev X How to X + X > C a...
-
What substitution products would you expect to obtain from the following reactions? (a) (c) Br T CH3CHCHCH3 + LII CHBr + NaCN ? (b) CH3 T CH3CHCHCI + HS ?
-
-Glucose has the following structure. Identify the chirality centers in -glucose, and tell how many stereoisomers of glucose are possible. CHOH - OH B-Glucose
-
Thun Company has been in operation for several years. It has both a deductible and a taxable temporary difference. At the beginning of 2019, its deferred tax asset was $690, and its deferred tax...
-
Mention and describe the varying levels of distribution intensity and the considerations in implementing each alternative. State the concept of vertical marketing systems and their application in...
-
user 9 A company produced in one month 16.000 units at total variable costs of P16,000. If 25,000 units are produced in the following month, the variable casts would be
-
The U.S. economy is starting to find its way through the economic turmoil caused by the pandemic. Government policy was very active with both monetary policy and fiscal policy attempting to keep the...
-
The volume can then be approximated with the Riemann sum A(x1)Ax++ A(xn)Ax, where n is the number of scans, Ax is the space between scans, and A(x) is the area of the organ given by the ith scan. For...
-
The total debt ratio is currently 69.57%. If the net income and total assets stay the same, but new shares are issued to pay off the debt, and the total debt ratio decreases to 50%, what would the...
-
Suppose that V is open in R2, that (a, b) V, and that f: V R is C3 on V. Prove that 2T limo 2. f(a + r cost, b+rsin ) cos(20) d = fxx(a , b) _fyy(a,b). r
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
Equation (3.38), C P = C V + TV ( 2 /). links C P and C V with and . Use this equation to evaluate C P C V for an ideal gas.
-
Draw the carbocation intermediate that would be formed if each of the following substrates participated in a stepwise elimination process (E1). In each case, identify the intermediate carbocation as...
-
Draw the transition state for the reaction between tertbutyl chloride and sodium hydroxide.
-
The following information pertains to ABC Corporation for the year ended December 31, 2023: Item Amount ($) Sales Revenue 11,000,000 Cost of Goods Sold 5,500,000 Gross Profit ? Operating Expenses...
-
Consider the following data for Burger King: Selling price per unit: $5 Variable cost per unit: $3 Fixed costs: $20,000 Complete the table below to calculate the break-even point and prepare a...
-
Determine the cost of goods sold if a company's beginning inventory was $30,000, purchases during the period amounted to $120,000, and the ending inventory was valued at $40,000. Discuss the...

Study smarter with the SolutionInn App


