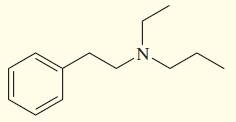
Show how you might synthesize the following tertiary amine three different ways, each using a different secondary
Question:
Show how you might synthesize the following tertiary amine three different ways, each using a different secondary amine and adding the final substituent by
(a) Reductive amination (3 ways).
(b) Acylation–reduction (3 ways).
Transcribed Image Text:
N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
aCondensation of the secondary amine with a carbonyl group ie an ald...View the full answer

Answered By

Thiruvengadam Vedamurthy
I have completed MPhil in Organic chemistry and MSc in chemistry. I am teaching for X, XI and XIIth CBSE as well as state board syllabus for more than two years. Currently , I am teaching chemistry for the NEET competitive examination.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how you might synthesize each of the following compounds using, as your starting materials, esters, ketones, acyl halides, and so on: (a) (b) (c) OEt OEt
-
Show how you might synthesize each of the following compounds from 1-butanol: (a) Butylamine (free of 28 and 38 amines) (b) Pentylamine (c) Propylamine (d) Butylmethylamine
-
Show how you might synthesize each of the following starting with a-tetralone (Section 15.9): (a) (b) (c) (d) HO So OH 02 H5
-
Bryce owns 200 shares of Basic Company stock that he purchased for $8,000 three years ago. On December 28, 2021, Bryce sold 100 shares of the stock for $2,500. On January 3, 2022, Bryce repurchased...
-
Explain why the total solubility of lead in Figure 6-3 first decreases and then increases as [ I-] increases. Give an example of the chemistry in each of the two domains.
-
If an object in stable equilibrium is displaced slightly, (a) there will be a restoring force or torque, (b) the object returns to its original equilibrium position, (c) its center of gravity still...
-
On March 15, 2017, Hunt Brands, a U.S. company, purchased merchandise from a South African company at a price of R1,000,000, payable in two months in rands. To hedge its exposed liability position,...
-
The Banisco Corporation is negotiating a contract to borrow $300,000 to be repaid in a lump sum at the end of nine years. Interest payments will be made on the loan at the end of each year. The...
-
If someone could help me with this question that would be amazing. I only want the direct method to be used. I REPEAT, I only want the direct method statement. If you could explain how you arrived at...
-
From the tenth floor of her office building, Katherine Rally watches the swarms of New Yorkers fight their way through the streets infested with yellow cabs and the sidewalks littered with hot dog...
-
A compound of formula C 11 H 16 N 2 gives the IR 1 H NMR, and 13 C NMR spectra shown. The proton NMR peak at 2.0 disappears on shaking with D 2 O Propose a structure for this compound, and show how...
-
Propose mechanisms for the nucleophilic acyl substitutions to form ethyl benzoate and N-methylacetamide as shown on the previous page.
-
California Instruments, Inc., produces 3,000 computer chips per day. Three hundred are tested for a period of 500 operating hours each. During the test, six failed: two after 50 hours, two at 100...
-
Explain how the marginal propensity to withdraw affects the outcome of a rise in autonomous expenditure.
-
How would the following transactions affect UK exports, imports and net exports? a. A British art lecturer spends the summer touring museums in Italy. b. Students in Paris flock to see the latest...
-
Which business cycle model do you find the most compelling and why?
-
What is the multiplier? Can the multiplier be negative as well as positive? Explain.
-
Suppose that survey measures of consumer confidence indicate a wave of pessimism is sweeping the country. If policymakers do nothing, what will happen to aggregate AD? What should the government do...
-
If there is a net loss from the sale of depreciable business property held long term, what is the character of the loss? How is it deducted (for or from AGI)?
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
Give the principal product(s) expected when 4-methyl-cyclohexene or other compound indicated reacts under the conditions in Problem 17.18. (a) Br2 in CH2Cl2, dark (b) A-bromosuccinimide in CCl4,...
-
Identify the benzylic carbons in the following structures. H,C-
-
A student Al Lillich has prepared a pure sample of 3-bromo-l-butene (A). Several weeks later he finds that the sample is contaminated with an isomer B formed by allylic rearrangement. (a) Give the...
-
Optimus Company manufactures a variety of tools and industrial equipment. The company operates through three divisions. Each division is an investment center. Operating data for the Home Division for...
-
(Prepared from a situation suggested by Professor John W. Hardy.) Lone Star Meat Packers is a major processor of beef and other meat products. The company has a large amount of T-bone steak on hand,...
-
Financial information for American Eagle is presented in Appendix A, and financial information for Buckle is presented in Appendix B. Required: 1. Calculate the cash holdings ratio for both companies...

Study smarter with the SolutionInn App


