Give the principal organic product(s) formed in each of the following reactions. (a) o-cresol + Br in
Question:
Give the principal organic product(s) formed in each of the following reactions.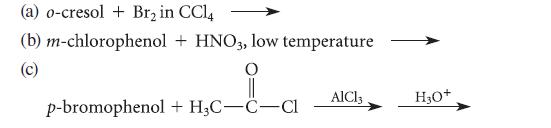
Transcribed Image Text:
(a) o-cresol + Br₂ in CCl4 (b) m-chlorophenol (c) + HNO3, low temperature || p-bromophenol + H₂C-C-Cl AlCl3 H₂O+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a OH Br ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the principal organic product of each of the following reactions: 2. H NaOH H-O 2. H 3. heat NaOCH.CH.ethanc 1. NaOH, H-O 2. H 3. heat NaOCH CHs. ethanol L. NaOH, HO 2. H 3....
-
Give the structure of the principal organic product of each of the following reactions: (a) (b) (c) (d) (e) COOCH2CH CH,CH2 heat COOCH CH COOCH,CH COOCH,CH NaOCH CH 2. H COOCH2CH3 Product of part (b)...
-
Give the structure of the principal organic product formed on reaction of benzyl bromide with each of the following reagents: (a) Sodium ethoxide (d) Sodium hydrogen sulfide (b) Potassium...
-
Explain how the structure of the balance sheet corresponds to the accounting equation.
-
A student on earth hears a tune on her radio that seems to be coming from a record that is being played too fast. She has a 33-rev/min record of that tune and determines that the tune sounds the same...
-
Calculate the deflections δB and δC at points B and C, respectively, of the cantilever beam ACB shown in the figure. Assume M0 = 36 k-in., P = 3.8 k, L = 8 ft, and EI = 2.25...
-
Internet companies have low fixed costs and low debt levels, yet their coefficients are high. Why?
-
The accounting records of Anderson Inc. show the following data for 2008. 1. Life insurance expense on officers was $9,000. 2. Equipment was acquired in early January for $200,000. Straight-line...
-
2 Rewrite cosx+ in terms of sin(x) and cos(x) 3
-
(a) Give the structure of the product formed in the reaction of urushiol with K 2 CO 3 and a large excess of methyl iodide. (b) Would this compound be likely to provoke the same allergic skin...
-
Electron transport takes place in the membrane of cellular organelles called mitochondria. What is it about the structure of ubiquinone and its reduction products that ensures their localization...
-
How much variation in voltage will most AC motors tolerate?
-
Acquiring versus Not Acquiring Alliance Partners As noted earlier, alliance partners with a high degree of network centrality benefit from being centrally located in a network of players. One debate...
-
What could be the key requirements that should be met with regard to procurement in South Africa?
-
Parker Plastic, Incorporated, manufactures plastic mats to use with rolling office chairs. Its standard cost information for last year follows: Standard Quantity Standard Price ( Rate ) Standard Unit...
-
The economy of South Africa has been in the doldrums for some time. Global uncertainty inadequate power supply and some structural constraints continue to hinder growth. These endogenous and...
-
(c) Barlucchi International Ltd is a multinational company based in the United Kingdom and manufactures beers, which are sold in two other countries namely, United States and France, through its...
-
Explain why the X-linked form of CGD is more common in boys and more severe than autosomal forms of the disease.
-
The figure shows a bolted lap joint that uses SAE grade 8 bolts. The members are made of cold-drawn AISI 1040 steel. Find the safe tensile shear load F that can be applied to this connection if the...
-
Which of the following cycloalkanes are capable of geometric (cis-trans) isomerism? Draw the cis and trans isomers. (a) 3-ethyl-1,1-dimethylcyclohexane (b) 1-ethyl-3-methylcycloheptane (c)...
-
Give IUPAC names for the following cycloalkanes. (a) (b) (c) CH CH,CH,CH, C(CH) CH,CH, CH H C
-
The heat of combustion of cis-1, 2-dimethylcyclopropane is larger than that of the trans isomer. Which isomer is more stable? Use drawings to explain this difference in stability.
-
An employee receives employer-provided adoption assistance. Which of the following are qualified adoption expenses can be used as an exclusion from income? Question 58Select one: a. Fees related to...
-
One of the flaws of GDP is that it Blank______. Multiple choice question. includes transactions of the underground economy accounts for changes in leisure and product quality accounts for...
-
What is a primary tax obligation for unincorporated NFPOs? Question 4 options: Non-Profit Organization Information Return (T1044) Corporation Income Tax Return (T2) GST/HST and payroll Registered...

Study smarter with the SolutionInn App


