Which of the two compounds in each of the following sets should react more rapidly in a
Question:
Which of the two compounds in each of the following sets should react more rapidly in a nucleophilic aromatic substitution reaction with CH3O¯ in CH3OH? Explain your answers.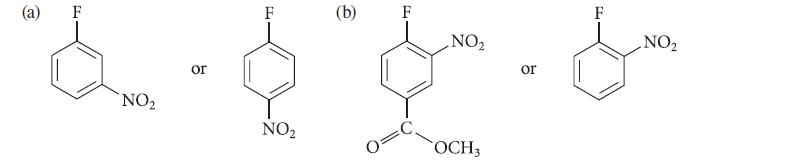
Transcribed Image Text:
(a) NO₂ or F NO₂ (b) NO₂ OCH3 or PL F NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The second compound pfluoronitrobenzene reacts mo...View the full answer

Answered By

Nishtha Goel
have more than 6 years of teaching experience in ICSE and CBSE School of Subject Maths, Science and English. Also I have done MSc Chemistry and MA English and a little experience of process Control and Instrumentation.
For a question to solve, you first have a basic knowlegde and Understanding of the subject which I will provide to you. Moreover to understand the topic discussion plays an important role, when we discuss a certain topic with someone many questions arise in our mind and to solve that questions, I am available for you.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain how you could distinguish between the two compounds in each of the following sets using only 13C NMR spectroscopy. Trans - 1,2-cy clohexanediami ne and trans - 1,4-cyclohexanediamine
-
Which of the two isomers in each of the following sets should have the greater basicity at the carbonyl oxygen? Explain. H,C CHCHC- OCH or HCC-o-CH CH CH
-
(a) The following resonance-stabilized ion can protonate to give two different constitutional isomers. Give their structures and give the curved-arrow notation for their formation. (b) One of the...
-
Consider a situation with two countries that have abatement cost functions : for j=L and j=H. The countries have identical damage functions D(E)= d.E.For each country the parameters s j are drawn...
-
Jay has been posted to a remote region of space to monitor traffic. Toward the end of a quiet shift, a spacecraft goes by, and he measures its length using a laser device, which reports a length of...
-
A steel column used in a college recreation center are 16.75 m long and are formed by welding three wide-flange sections (see figure). The columns are pin-supported at the ends and may buckle in any...
-
What is the drawback of company rankings based on EVA?
-
CVP analysis is both simple and simplistic if you want realistic analysis to underpin your decisions, look beyond CVP analysis. Do you agree? Explain.
-
Problems 4-7: Simplify each rational expression. If the rational expression cannot be simplified, so state. 48y5 4) 15y3 4a-9b2 5) 2a-3b 6) 3x+1 3x-29x-10 3-x 7) Sr-15
-
Calculate the oxidation state of the metal in each of the following complexes. (a) O Mn-O- permanganate (b) Pd(PPH3)4 tetrakis(triphenylphosphine) palladium (c) CpFe ferrocene
-
Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both L-type and X-type bonds. Use M as a general metal. TABLE 18.1 Some Typical Ligands Used in...
-
How are opportunity costs different from other costs mentioned in the chapter? Can you see a problem with using opportunity costs when making management decisions?
-
Our new business venture of creating a comfortable, luxurious, and modern office spaces accessible for the average person.A direct competitor would be WeWork. Discuss how they are a direct...
-
Segmentation is key to identifying the right target markets. As a start-up company, SOCIAL LITE Vodka knew it had to be extremely focused on segmentation. Its alcoholic beverages were made with 100...
-
Jay Aquire is considering the purchase of the following : a Builtrite , $ 1000 par , 6 5 / 8 % coupon rate , 25 year maturity bond which is currently selling for $ 1020 . If Jay purchases this bond...
-
a) Examine FIVE (5) technology and business trends that have enhanced the role of information systems in today's competitive business environment. (15 Marks) b) Assess the capabilities of a digital...
-
Several accounts and amounts from the financial statements of a recently opened Big Poppy Coffee appear below for the year ending December 31, Year 1. Equipment 43,600 Cash $ 10,600 Note payable, due...
-
Devise an experiment that will differentiate between bactericidal and bacteriostatic effects.
-
Let X be a random variable taking on values a1, a2, . . . , pr with probabilities p1, p2, . . . , pr and with E(X) = μ. Define the spread of X as follows: This, like the standard deviation, is a...
-
For each reaction, estimate whether ÎSo for the reaction is positive, negative, or impossible to predict. a. (b) The formation of diacetone alcohol: c. heat C10H22--> n-decane C3H6 + C7H16...
-
(a) Propose a mechanism for the free-radical chlorination of ethane, (b) Calculate ÎHo for each step in this reaction. (c) Calculate the overall value of ÎHo for this reaction. hu CH3...
-
Draw Lewis structures for the following free radicals. (a) The ethyl radical, (b) The tert-butyl radical, (CH3)3C (c) The isopropyl radical (2-propyl radical) (d) The iodine atom CH3 CH2
-
ABC Corp., a US corporation, purchased goods on credit from a British company on April 8, 2007. ABC made a payment of 10,000FC on May 8, 2007. The exchange rate was $1= FC .50 on April 8 and $1= FC...
-
Which of the following groups should be the focal point of a company's strategy? MulABC Inc. produces a single product, In doing so, the company incurs overhead costs as follows: Indirect Factory...
-
When capturing and documenting system audit requirements for an IT project, the teams is focused on capturing the following

Study smarter with the SolutionInn App


