Within each series, arrange the compounds according to increasing rates of their reactions by the S N
Question:
Within each series, arrange the compounds according to increasing rates of their reactions by the SN1–E1 mechanism. Explain your reasoning.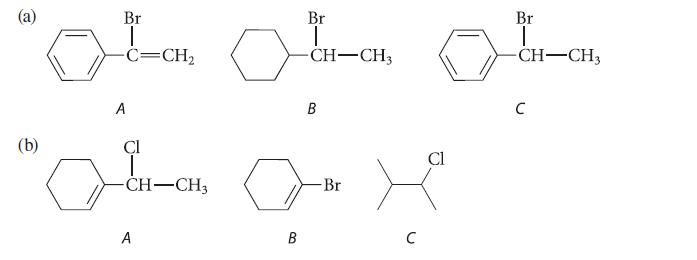
Transcribed Image Text:
(b) Br -C=CH, A Cl امي مسلم -CH-CH3 A Br | -CH-CH3 B B Br لى C Cl Br -CH-CH3 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b The o...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Within series, arrange the compounds according to increasing rates of their reactions by the SN1 - E1 mechanism. Explain your reasoning. CH CH Br
-
Within each series arrange the compounds in order of increasing stability: C(CH3)3 HO CH,CH(CH)
-
Within set, rank the compounds in order of increasing rates of their SN2 reactions. Explain your reasoning. 1-bromocyclohexene, bromocyclohexane, 1-(bromomethyl)cyclohexene
-
The example images in the text for Fade do not quite line up in the vertical direction (the mandrill's mouth is much lower than Darwin's). Modify Fade to add a transformation in the vertical...
-
The proper mean lifetime of a muon is 2 s. Muons in a beam are traveling at 0.999c. (a) What is their mean lifetime as measured in the laboratory? (b) How far do they travel, on average, before they...
-
An aluminum pipe column (alloy 2014-T6) with pinned ends has outside diameter d2 = 5.60 in. and inside diameter d1 = 4.80 in. (see figure). Determine the allowable axial load Pallow for each of the...
-
If EPS rises after a deal, does this necessarily imply value creation?
-
Some fast-food chains offer a lower-priced combination meal in an effort to attract budget-conscious customers. One chain test-marketed a burger fries and a drink combination for $1.71. The weekly...
-
Consider the following function. f(x) = x-8x+7 (a) Find the critical numbers of f, if any. (Enter your answers as a comma-separated list.) x= (b) Find the open intervals on which the function is...
-
Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both L-type and X-type bonds. Use M as a general metal. TABLE 18.1 Some Typical Ligands Used in...
-
Within each set, rank the compounds in order of increasing rates of their S N 2 reactions. Explain your reasoning. (a) Benzyl bromide, (3bromopropyl) benzene, pbromotoluene (b) 1bromocyclohexene,...
-
The Catherine wheel is a firework that consists of a coiled tube of powder which is pinned at its center. If the powder burns at a constant rate of 20 g/s such that the exhaust gases always exert a...
-
Flint Corp. acquired a property on September 15, 2023, for $230,000, paying $3,500 in transfer taxes and a $1,700 real estate fee. Based on the provincial assessment information, 80% of the...
-
A. What are some of the main ideas (minimum two) that stood out to you from the video https://www.ted.com/talks/amy_edmondson_how_to_turn_a_group_of_strangers_into_a_team How to turn a group of...
-
Profits have been decreasing for several years at Pegasus Airlines. In an effort to improve the company's performance, the company is thinking about dropping several flights that appear to be...
-
Analyze a recent business crisis and evaluate the communication strategies employed by the organization in response to the crisis. What lessons can be learned about the impact of timely, transparent,...
-
Conduct investigation into current trends in healthcare using the BU Library search capabilities and any other materials/resources provided to you in the course. Discuss what you think are some of...
-
It may seem contradictory that lyophilization can be used to preserve foods yet also be used to keep cultures of microbes viable. Explain how these applications can be reconciled.
-
Which provision could best be justified as encouraging small business? a. Ordinary loss allowed on $ 1244 stuck. b. Percentage depletion. c. Domestic production activates deductions. d. Interest...
-
When a small piece of platinum is added to a mixture of ethene and hydrogen, the following reaction occurs: Doubling the concentration of hydrogen has no effect on the reaction rate. Doubling the...
-
(a) Draw the reaction-energy diagram for the reverse reaction: CH3 + HCl CH4 + Cl (b) What is the activation energy for this reverse reaction? (c) What is the heat of reaction (H) for this reverse...
-
Draw a reaction-energy diagram for the following reaction: CH3 + Cl2 CH3Cl + Cl The activation energy is 4 kJ mol (1 kcal mol), and the overall Ho for the reaction is -109kJ/mole (-26 kcal/mol) (b)...
-
T4 information slips and T4 summary forms must be submitted by: Multiple Choice The final day of February of the following year A convenient date determined by the employer 30 days after the year-end...
-
During 2021/22 Tax Year, Selma Car (aged 63), received a superannuation income stream benefit of $57,200 from her superannuation funds. As her superannuation was derived from an untaxed element...
-
COMPREHENSIVE PROBLEM 2 - The following ledger accounts appear below, in this order: Cash, Accounts Receivable, Inventory, Estimated Returns Inventory, Prepaid Insurance, ...

Study smarter with the SolutionInn App


