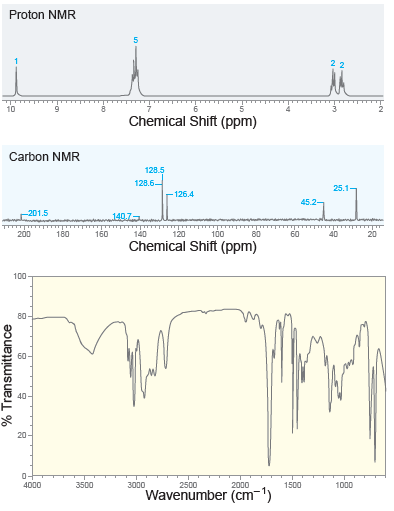
A compound with molecular formula C 9 H 10 O exhibits the following spectra ( 1 H
Question:
Transcribed Image Text:
Proton NMR 10 Chemical Shift (ppm) Carbon NMR 128.5 128.6- 25.1- 126.4 201.5 140.7 160 140 120 100 200 180 80 60 40 20 Chemical Shift (ppm) 100 80 - 60- 40- 20- 0- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-') % Transmittance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound with molecular formula C 9 H 10 O exhibits a strong signal at 1687 cm 1 in its IR spectrum. The 1 H and 13 C NMR spectra for this compound are shown below. Identify the structure of this...
-
Identify the compound with molecular formula C8H10O that gives the IR and 1H NMR spectra shown in Figure 14.23. 23 16 27 2 29 35 13 14 15 16 800 200 2400 200 0 6 (ppm) frequency
-
Propose a structure for a compound with molecular formula C 3 H 8 O that exhibits the following 1 H NMR and 13C NMR spectra: Proton NMR 0.5 5.0 4.5 4.0 3.5 3.0 2.0 1.5 1.0 25 Chemical shift (ppm)...
-
Every real number is either a/an number or a/an_______ number.
-
Explain the differences among the three major forms of bankruptcy: Chapter 7, Chapter 11, and Chapter 13.
-
A spring gun (k = 28 N/m) is used to shoot a 56-g ball horizontally. Initially the spring is compressed by 18 cm. The ball loses contact with the spring and leaves the gun when the spring is still...
-
Exron Oil and Gas Company constructs a natural gas treatment facility in three phases. The first phase was completed and placed into service on December 31, 2017. The second phase was completed and...
-
Cash CollectionsDirect Method Emily Enterprises comparative balance sheets included accounts receivable of $224,600 at December 31, 2009, and $205,700 at December 31, 2010. Sales reported on Emilys...
-
Sales Joel de Paris, Incorporated Income Statement Operating expenses Net operating income Interest and taxes: Interest expense Tax expense Net income $ 4,850,000 3,977,000 873,000 $110,000 202,000...
-
Melcher Enterprises is a wholesaler that purchases consumer merchandise from many different suppliers. Melcher then sells this merchandise to many different retail chain stores. The following...
-
Solid methanol in thermal contact with the surroundings is reversibly melted at the normal melting point at a pressure of 1 atm. Are S, S surroundings , and S total positive, negative, or zero?...
-
Can incandescent lighting be regarded as an example of cogeneration during the heating season? In a season where air conditioning is required?
-
It is not possible to add a new window type to a window family. A) True B) False
-
Cadott Manufacturing produces three products. Production and cost information show the following: Model X Model Y Model Z Units produced 1,000 3,000 6,000 Direct labor hours 2,000 1,000 2,000 Number...
-
Evaluate each limit, if it exists. Hint: use Rationalization. a. lim x-0 8 9+x - 3 b. lim x-0 3 x x + 3 - x
-
XYZ has $25,000 of debt outstanding and a book value of equity of $25,000. The company has 10,000 shares outstanding and a stock price of $10. If the unlevered beta is 0.75 and the marginal tax rate...
-
Problem 01.008 - Stress in a link supporting a conveying system Link AC has a uniform rectangular cross section in. thick and 1 in. wide. Determine the normal stress in the central portion of the...
-
As an operations manager of the hotel, how much he or she can expect to gain in additional quality (percentage wise of the total complaints in the sample) by generally addressing the bottom three...
-
Define the internal path length, I(T), of a tree T to be the sum of the depths of all the internal nodes in T. Likewise, define the external path length, E(T), of a tree T to be the sum of the depths...
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
We can conceive of a stepwise version of the SN2 reaction consisting of a Lawis acid-base dissociation followed by a Lewis acid-base association. (Nuc:- = a nucleophile.) (a) Why should the stepwise...
-
We can conceive of a stepwise version of the SN2 reaction consisting of a Lawis acid-base dissociation followed by a Lewis acid-base association. (Nuc:- = a nucleophile.) (a) Why should the stepwise...
-
I need the answer please Question: pf1-exam-npc-g-f Step 1-Calculate Mara's gross earnings | Mara Poirier works for Affordable Transport in Quebec and earns an annualsalary of $47,500.00 paid on a...
-
Scenario 3 - Sally McCallan Sally just turned 20 and has saved $10,000 as a deposit for a property. She is a First Home Buyer but does not qualify for the First Home Owner Grant as the property is...
-
Based on this information: Multiple Choice Both direct materials and rent expense are variable costs. Direct materials is a fixed cost and rent expense is a variable cost. Both direct materials and...

Study smarter with the SolutionInn App


