Compare the structures of ethylene, acetylene, and benzene. Each of these compounds produces only one signal in
Question:
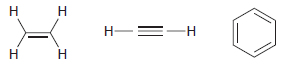
Transcribed Image Text:
Н Н Н- -н Н H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 22% (9 reviews)
Increasing chemical shift in ...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Compare the structures of a synchondrosis and a symphysis.
-
Compare the structures of HNO2 and H2CO3. Which would you expect to be the stronger acid? Explain your choice.
-
Compare the structures of 1,4-pentadiene and divinyl amine: The first compound does not absorb UV light in the region between 200 and 400 nm. The second compound does absorb light above 200 nm. Using...
-
Starting with acetylene as your only source of carbon atoms, identify how you would prepare each member of the following homologous series of aldehydes: a. Ethanal b. Propanal c. Butanal d. Pentanal
-
What are three examples of popular messaging systems?
-
Richardson Manufacturing Company is considering purchasing a machine that will cost $ 180,000. The machine is expected to have a useful life of 8 years, with no expected salvage value ; however, for...
-
Washington State Products has several manufacturing divisions. The Seattle Division produces a component part that is used in the manufacture of electronic equipment. The cost per part for July is as...
-
Classic Automobiles of Cedar Grove, Inc., was formed on January 1, 2012. The following transactions occurred during 2012: On January 1, 2012, Classic issued its common stock for $430,000. Early in...
-
History and revolution of taxation Current ordanance or act of taxation will be used for taxation system Types of taxation Income from salary tax
-
As chairman of the board of ASP Industries, you estimate that your annual profit is given by the table below. Profit (() is conditional upon market demand and the effort of your new CEO. The...
-
How many signals will be expected in the 1 H NMR spectrum of each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) `NO2
-
The 2009 balance sheet and income statement for the Woods Company are shown here: Woods Company: Income Statement for the Year Ending December 31, 2009 ($ thousands)...
-
In divisional income statements prepared for Wilborne Construction Company, the Payroll Department costs are charged back to user divisions on the basis of the number of payroll checks, and the...
-
A company would like to pick the best option out of the two described in the table below. The table shows the net cash flow for each year of the 10 year planning horizon. MARR is 12% EOY Option 0...
-
The yield to maturity for a company's long-term debt is 8.1% per year. The company's common stock has a market value of $47.89 per share and its last dividend was $3.15 per share. The stock's beta is...
-
Question 18 (1 point) Saved Tesin(x) x < 0 f(x)=e+sin(x) 0 < x < e - sin(x) X > 18. (1 mark) Given f(x) above, the values of lim f(x) and f(T) respectively are: a b C d e a) DNE, T b) e", e HT c) e,...
-
Manila Company manufactures leather bags and uses the job order cost system. Its work in process show: Direct materials used P341,000 Direct labor cost 324,500 Factory overhead 259,600 Transferred to...
-
K A company issues a 100-day, 12% note for $16,000. What is the principal amount of the note? (Round your answer to the nearest dollar.) OA. $16,000 OB. $17,920 OC. $16,533 OD. $15,467
-
Refer to Exercise 80. Would you be surprised if the train arrived on time on fewer than 4 days? Calculate an appropriate probability to support your answer. Exercise 80. According to New Jersey...
-
Do public and private companies follow the same set of accounting rules? Explain.
-
How would you carry out the following transformations? More than one step is required in each case. (a) H (b) Ph "Co "
-
The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxyl group in the Meta position increases the acidity. . CO2H CO2H...
-
3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odor of human sweat. Synthesize the compound from starting materials having five or...
-
Which of the following is not a responsibility of a Credit Reporting Agency under the FCRA? You Answered Requesting that a data furnisher investigate and verify disputed account information Providing...
-
A landlord is paid $82,000 by its commercial tenant as a lease cancellation fee. The tenant wanted to get our ot its lease so they could move to a different city. The landlord had held the lease for...
-
Which organization's values are most likely to be effective? Select a Choice Below current question choices OptionA Alpha Corp's values were created based on the inputs from all employees. OptionB...

Study smarter with the SolutionInn App


