Cyclopropane (shown below) has only one constitutional isomer. Draw a condensed structure of that isomer. -.
Question:
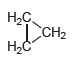
Transcribed Image Text:
Н-С. Т Сн- CH2 H2Č
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
H ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propylene is produced by cracking petroleum and is a very useful precursor in the production of many useful polymers. Propylene has one constitutional isomer. Draw that isomer, and identify its...
-
Two constitutional isomers having molecular formula C4H6O are both symmetrical in structure. In their infrared spectra, neither isomer when in dilute solution in CCl4 (used because it is nonpolar)...
-
Vaccenic acid, a constitutional isomer of oleic acid, has been synthesized through the following reaction sequence: Propose a structure for vaccenic acid and for the intermediates A-E. liq. NaUN KUH,...
-
Your input file contains sentences. A sentence contains words seperated with one space or more spaces. And a sentence ends with a "." Symbol. Write appropriate lex code to create desired outputs....
-
What is the role of arbitragers in futures markets? What impact do they have on futures prices?
-
Show how the IS and LM schedules look in the monetarist view. Use these schedules to illustrate the monetarist conclusions about the relative effectiveness of monetary and fiscal policy.
-
The two accounts below were taken from the books of a retailer at the end of his financial year, 31 December 19X7. Required: Answers to the following questions. Insurance Account Dr 19X7 19X7 Jan 1...
-
The ledger of Montgomery Company at the end of the current year shows Accounts Receivable $78,000; Credit Sales $810,000; and Sales Returns and Allowances $40,000. Instructions (a) If Montgomery uses...
-
Please show step by step and formulas used A.The ER is real/$ .10, or $10 buys 1 real. The change in the ER is what percentage wise, if at the end of the year the ER is r/$ .011111111, or $ 90 bu...
-
The synchronous machine of Problem 4.9 has a three-phase winding with 45 series turns per phase and a winding factor kw = 0.928. For the flux condition and rated speed of Problem 4.9, calculate the...
-
Propose a molecular formula for a compound that exhibits the following peaks in its mass spectrum. a) (M) + at m/z = 72, relative height = 38.3% of base peak (M+1) + at m/z = 73, relative height =...
-
Most common amines (RNH 2 ) exhibit pK a values between 35 and 45. R represents the rest of the compound (generally carbon and hydrogen atoms). However, when R is a cyano group, the pK a is found to...
-
Consider a cube of mass m and side a. (a) Show that the ellipsoid of inertia at the center of the cube is a sphere, and use this property to determine the mass moment of inertia of the cube with...
-
How are the work in process inventories reported in the cost of goods manufactured schedule?
-
What do we mean when we say things can communicate with each other? What types of communication can they undertake?
-
Is it possible for every single thing (living or nonliving) to be connected to the internet (e.g. could dust, water, bacteria, diseases, plates, trees, volcanoes, etc. be connected to the internet)?
-
What is a cyber-physical System (CPS)? Are there different types of cyber-physical systems?
-
Identify and discuss the three broad functions of management.
-
Use the trace route (UNIX) or tracert (Windows) programs to trace the route from your computer to various universities on other continents. Make a list of transoceanic links you have discovered. Some...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
The second step in the strategic-management process is to look at where the organization stands and see what is working and what could be different to maximize efficiency and effectiveness in...
-
The ending balance of the retained earnings account appears in a.both the statement of stockholders' equity and the balance sheet b.both the statement of stockholders' equity and the statement of...
-
The following items are taken from the financial statements of Tracy Company for 2022. Tracey Company will prepare a balance sheet at December 31, 2022 and an income statement from January 1, 2022 to...

Study smarter with the SolutionInn App


