Deduce the structure of a compound with molecular formula C 8 H 10 O that exhibits the
Question:
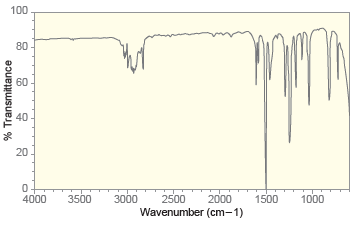
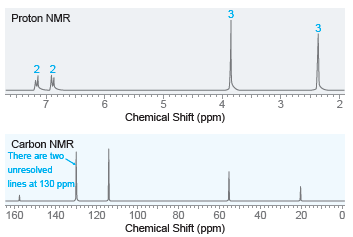
Transcribed Image Text:
100 80 60 40 20 2500 2000 Wavenumber (cm-1) 1000 4000 3500 3000 1500 % Transmittance 3 Proton NMR 3 2 2 Chemical Shift (ppm) Carbon NMR There are two, unresolved lines at 130 ppm 160 140 120 100 80 60 40 20 Chemical Shift (ppm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 3000 2500 Wavenumber (cm-1) 4000 3500 2000 1500...
-
Deduce the structure of a compound with molecular formula C 6 H 14 O 2 that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. 100 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 Wavenumber...
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
public class Class extends ClassA public classB ( ( Time left 0 0 : 1 2 : 0 3 int init = 1 0 ; super ( 4 0 ) ; O a . . The method super is not defined. b . . No values may be passed to super. O c . ....
-
What are three advantages of telecommuting?
-
An electric motor under steady load draw s 9.7 amperes at 110 volts, delivering 1.25(hp) of mechanical energy, what is the rate of heat transfer from the motor, in kW?
-
Suppose a firm has a book balance of \($2\) million. At the automatic teller machine (ATM), the cash manager finds out that the bank balance is \($2.5\) million. What is the situation here? If this...
-
Comprehensive Problem. As of July 1, 2011, the trial balance for Korner College was as follows: During the year ended June 30, 2012, the following transactions occurred: 1. Cash collections included:...
-
You invest $1,000 in a large company stock and $1,000 in a corporate bond. If you earn 10.0 percent on the stock and 6.0 percent on the bond and hold each security for 10 years, what are the terminal...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Look back at Figure 2, which shows the inverse relationship between ticket prices and game attendance at Gigantic State University. (a) Interpret the meaning of both the slope and the intercept. (b)...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Use a CAS to perform the following steps. a. Plot the space curve traced out by the position vector r. b. Find the components of the velocity vector dr / dt. c. Evaluate dr / dt at the given point t...
-
a. What objectives are met in obtaining a client representation letter? b. What is the impact of a client's refusal to provide a rep letter.
-
What was Tootsie Roll's largest current asset, largest current liability, and largest item under "Other assets" at December 31, 2007 ?
-
a. Identify the four types of attest engagements that have been recognized in professional standards. b. Indicate the levels of assurance and attestation risk associated with each type.
-
a. Distinguish three situations involving information accompanying audited financial statements. b. Describe the auditor's responsibilities for each type of information.
-
a. How are the attestation standards classified? b. Indicate the principal differences between the attestation standards and GAAS.
-
The splay tree does not have good performance for the sorted map operations, because those methods lack calls to the rebalanceAccess hook. Reimplement TreeMap to include such calls.
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Draw structures corresponding to the following systematic names: (a) (4E)-2, 4-Dimethyl- 1, 4-hexadiene (b) cis-3, 3-Dimethyl-4-propyl-1, 5-octadicne (c) 4-Methyl-1, 2-pentadiene (d) (3E, 5Z)-2,...
-
Menthene, a hydrocarbon found in mint plants, has the systematic name 1-isopropyl-4-methylcyclohexene. Draw its structure.
-
Draw and name the 6 pentene isomers, C5H10, including E, Z isomers.
-
. Han Company has three products in its ending inventory. Specific per unit data at the end of the year for each of the products are as follows: Product 1 Product 2 Product 3 Cost $ 40 $ 110 $ 70...
-
. Swifty Corporation is projecting a cash balance of $24.600 in its December 31. 2024. balance sheet. Swifty's schedule of expected collections from customers for the ?rst quarter of 2025 shows ...
-
Find the k-best (i.e. largest) values in a set of data. Assume you are given a sequence of values, one value at a time. We do not know how many elements there are in this sequence. In fact, there...

Study smarter with the SolutionInn App


