Draw all significant resonance structures for each of the following compounds: a. b. c. d. e. f.
Question:
a.
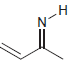
b.
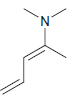
c.
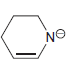
d.
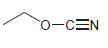
e.
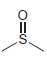
f.
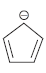
g.
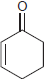
h.
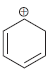
i.
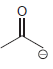
j.
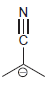
k.
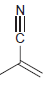
l.
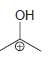
Transcribed Image Text:
.Н z: N.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a b c ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw all significant resonance structures for each of the following compounds: Testosterone Estradiol (Female sex hormone) (Male sex hormone)
-
Draw contributing resonance structures for each of the following species, and rank the structures in order of decreasing contribution to the hybrid: a. b. c. d. e. f. CH3C-CH CHCH3 CH3 0 CH3COCH3 +OH...
-
Draw resonance structures for each of the following radicals: (a) (b) (c) (d)
-
Besides warehouse layout decisions, what are some other applications where ranking items according to bang/buck might make sense?
-
Vogts Company sells TVs. The perpetual inventory was stated as $38,500 on the books at December 31, 2014. At the close of the year, a new approach for compiling inventory was used and apparently a...
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
Yale Corporation issued to Zap Corporation \(\$ 60,000,8 \%\) (cash interest payable semiannually on June 30 and December 31) 10 -year bonds dated and sold on January 1, 2020. Assume that the company...
-
Dave Fletcher was able to deter-mine the activity times for constructing his laser scanning machine. Fletcher would like to determine ES, EF, LS, LF, and slack for each activity. The total project...
-
7. Valuation of interest rate swap A $100 million interest rate swap has a remaining life of 22 months. Under the terms of the swap, 6- month LIBOR is exchanged for 6% per annum (compounded sem...
-
Irene Pypypiv has been working as a dishwasher at a bakery, while attending pastry arts school. As a graduation present, Irenes parents give her a graduation present of $40,000, which matches the...
-
Draw resonance structures for each of the following compounds: a. b. c. d. e. f. g. h. i. j. OH
-
A sealed flask with a capacity of 1.22 dm 3 contains 4.50 g of carbon dioxide. The flask is so weak that it will burst if the pressure exceeds 9.500 10 5 Pa. At what temperature will the pressure of...
-
Without performing any calculations, determine which measure of central tendency best represents the graphed data. Explain your reasoning. Body Mass Indexes (BMI) of People in a Gym 18 20 22 24 26 28...
-
A firm's current balance sheet is as follows: Assets $ 120 Debt $ 12 Equity $ 108 A. What is the firm's weighted-average cost of capital at various combinations of debt and equity, given the...
-
3) What is the Capacitance of a parallel-plate capacitor made up of two aluminum plates, 4 cm in length on a side and separated by 5 mm? 4) If 10J of work is required to move 2C of charge in a...
-
A regular hexagon with side length 6 units is rotated in 3D space to form a solid. Calculate the surface area of the resulting shape.
-
7.15 Assume that X1, X2, X3, X4 are independent and normally distributed with E[X] = 100 E[X2] = 90 E[X3] = 95 E[X4]=105 Var[X1] = 30 Var[X2] = 20 Var[X3] = 25 Var[X4]=15. (a) What is the...
-
The Koch snowflake is a fractal formed by repeatedly adding smaller equilateral triangles to the sides of an initial triangle. If the perimeter of the initial equilateral triangle is 36 units,...
-
Randomly selected subjects are inflicted with tinea pedis (athlete?s foot) on each of their feet. One foot is treated with a fungicide solution while the other foot is given a placebo. The results...
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
Write structural formulas for the products that would be formed when geranial, a component of lemongrass oil, is treated with ozone and then with dimethyl sulfide (Me2S). Geranial
-
Limonene is a compound found in orange oil and lemon oil. When limonene is treated with excess hydrogen and a platinum catalyst, the product of the reaction is 1-isopropyl-4-methylcyclohexane. When...
-
Pheromones (Section 4.7) are substances secreted by animals that produce a specific behavioral response in other members of the same species. Pheromones are effective at very low concentrations and...
-
Which represent a formula or equation for break-even in units? (check all that apply) Fixed Costs/Contribution Margin Ratio Fixed Cost / Contribution Margin per Unit 0 = Contribution Margin per Unit...
-
Chapter 3 Income Statements (P&Ls) Chapter 3 Income Statements (P&Ls) Use Richter's steak house p &l for April 2012. Add a column to the report for percentages of each category. Show profitability if...
-
2. Chapter 2 - Net Asset Acquisition (Ch 2 in class ex, text 45) Donner Corporation had the following account balances and fair values at December 31, 2023 when the net assets were acquired by...

Study smarter with the SolutionInn App


