Draw the mechanism of the following reaction, and make sure to draw all three resonance structures of
Question:
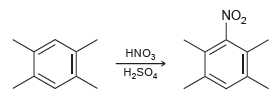
Transcribed Image Text:
NO, HNO3 H,SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
28 H oja OTH H ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the mechanism of the following reaction. This reaction is the reverse of sulfonation, so you should read the sulfonation mechanism backward. Your mechanism should involve a sigma complex...
-
Draw the mechanism of the following reaction, which involves two consecutive Friedel-Crafts alkylations. When drawing the mechanism, do not try to draw the two alkylations as occurring simultaneously...
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
Karamazov Semiconductors is considering an investment to expand its existing line of business. The investment will cost $10 million and is expected to produce after-tax cash flows of $1 million per...
-
Visit the Web site of the National Institute of Standards and Technology (www.nist.gov/baldrige/), the organization that grants the Malcolm Baldrige National Quality Award, the highest quality award...
-
An astronaut wants to remove a bolt from a satellite in orbit. He positions himself so that he is at rest with respect to the satellite, then pulls out a wrench and attempts to remove the bolt. What...
-
During 2015, Post Oil and Gas Company completed the last well from its drilling and production platform off the coast of Louisiana. Unrecovered costs not including decommissioning costs on December...
-
The following data were taken from the general ledger and other data of McDonough Manufacturing on July 31: Finished goods, July 1................................................... $ 85,000 Cost of...
-
If y=2x ln(x), find the following. y=0
-
For each of the following transactions of Vickys Day Care Center, identify the accounts that would be increased and those that would be decreased: 1. The center borrowed $ 10,000 from a bank. 2. Rent...
-
Calculate KP at 298 and 490. K for the reaction NO(g) + 1/2O 2 (g) NO 2 (g) assuming that H o R is constant over the interval 298600. K. Do you expect K p to increase or decrease as the temperature...
-
A sample containing 2.75 moles of N 2 and 6.25 mol of H 2 is placed in a reaction vessel and brought to equilibrium at 52.0 bar and 690. K in the reaction 1/2N 2 (g) + 3/2H 2 (g) NH 3 (g). a....
-
Give two purposes to controls over batch report collection. Briefly describe two controls that can be established to ensure these objectives are achieved.
-
Draw, and carefully label the graphs listed below for a perfectly competitive industry. Hint: draw graph 1 on the left, and graph 2 on the right. . Graph 1: Market supply and demand. . Graph 2:...
-
Earnie sells lemonade at a busy street corner in Rollaville. His production function is q-f(x,y)=x1/41/2 where output is measured in gallons, x is the number of pounds of lemons he uses, and y is the...
-
III. Given a directed graph G = (V,E),where V = {v1, v2, v3, v4, v5, v6}, and E = {(v1, v2), (v1, v3), (v1, v4), (v2, v5), (v3, v5), (v4, v6), (v5, v4), (v5, v6)}: 1. give the adjacency matrix for G...
-
Complete the following calculations taking into account correct significant figrues pH of diluted solution H* concentration in diluted solution H* concentration in concentrated solution OH...
-
2) Consider air flow (at the film temperature, p=1.2 kg/m and u=2x105 Pa.s, k=0.026 W/m.K, Pr=0.7) around a cylinder with diameter of XX cm. The cylinder surface temperature is 10*XX more than the...
-
What is lot sizing and what is its goal?
-
Assume today is the 21st of February. Using the information below, FT Extract, answer the following questions (parts i and ii). You work for a US company that is due to receive 250 million in June...
-
In a laboratory, two liquids, A and B, were found in a box labeled only "isomeric alkyl halides C5H11Br." You have been employed to deduce the structures of these compounds from the following data...
-
What is the difference in the standard free energies of activation at 25oC of two reactions A and B if reaction B is 450 times faster than reaction A ? Which reaction has the greater G?
-
Which one of the following stereoisomers should undergo β-elimination most rapidly with sodium ethoxide in ethanol? Explain your reasoning. CH1 CH3 Br CH3 CH
-
Barnett Brass sells economy door knobs for $15 each. Unit product costs are as follows: Direct materials Direct labor Manufacturing overhead Total $ 3 2 6 $11 An order to purchase 4,000 units was...
-
Culver Inc. presented the following data. Net income $2,610,000 Preferred stock: 51,000 shares outstanding, $100 par, 7% cumulative, not convertible 5,100,000 Common stock: Shares outstanding 1/1...
-
Which of the following comments is true regarding Justin Paperny's comments during his presentation? A. Justin believes that the main reason he was sentenced to prison is because his actions were key...

Study smarter with the SolutionInn App


