Identify which of the following monomers would be most reactive toward anionic polymerization: CH3 OAc CN
Question:
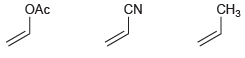
Transcribed Image Text:
CH3 OAc CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 65% (20 reviews)
least r...View the full answer

Answered By

Ashish Buragohain
For me tutoring is very good.It is a learning curve for me as well as for students.Through my time in tutoring i have learn many things which helped me in my profession.I have also learnt that not only we have to be good at explaining but also at giving encouragement and motivating learners.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify which of the following monomers would be most reactive toward cationic polymerization. OAc CN CI
-
Which of the following monomers might you expect would lead to a conducting polymer? a. b. H2C==CHCH3 --,
-
Which of the following monomers might you expect to lead to a conducting polymer? a. CH3CH==CHCH3 b. CH CC CH
-
(a) the maximum shear stress magnitude T1 in aluminum tube (1). (b) the maximum shear stress magnitude T2 in brass shaft segment (2). (c) the maximum shear stress magnitude T3 in brass shaft segment...
-
Consider the following partially completed income statements for merchandising companies and compute the missing amounts: Smith, Inc. Allen, Inc. $ 101,000 Net Sales Revenue (d) Cost of Goods Sold:...
-
Raj Kothare funded an irrevocable simple trust in May of last year. The trust benefits Raj's son for life and grandson upon the son's death. One of the assets he transferred to the trust was Webbco...
-
Eugene Incorporated had the following transactions involving current assets and current liabilities during February 2002. Additional information: 1. As of February 1, 2002, current assets were...
-
Assume that your organization is planning to have a server room that functions without human beingsin other words, the functions are automated (such a room is often called a lights-out server room)....
-
On January 1 of this year, Cunningham Corporation issued bonds with a face value of $200,000 and a coupon rate of 6 percent. The bonds mature in 10 years and pay interest annually every December 31....
-
The company estimates that its cost of capital is 14%. Calculate the payback period for Machine A and B (answers must be expressed in years, months and days). 2.1 (5 marks) Calculate the accounting...
-
Nitroethylene undergoes anionic polymerization so rapidly that it is difficult to isolate nitroethylene without it polymerizing. Explain.
-
Draw a region of an alternating copolymer constructed from vinyl chloride and ethylene.
-
What is meant by antecedent conditions? How do they shape the value of objects?
-
what ways do organizational justice and perceived fairness impact employee engagement, motivation, and retention in a diverse workforce ?
-
2. Say (X,d) is a metric space and A is a nonempty subset of X. Define a real-valued function f(p) on X by (p) = inf{d(p. x)|2 A}. a) Show that f(p) f(g)| d(p. q) b) Use a) to show that is a...
-
You live in a city where the atmospheric pressure is either101.3 kPa (or else 14.69 psi). If you take a vessel andpressurise it to 350 kPa (or 50.75 psi) above the atmosphericpressure and connect...
-
Information for two companies follows: Skittles Company Sales $ 6,151,450 Contribution margin Fixed costs 4,951,450 3,897,950 Starburst Company $ 3,739,000 1,414,000 909,000 (1) Compute the degree of...
-
Write a program that will ask the user to input the number of dice to roll, and the number of sides on the dice. It will then roll the dice and show the results of each roll as well as the total of...
-
Write HDL code for the multicycle MIPS processor. The processor should be compatible with the following top-level module. The mem module is used to hold both instructions and data. Test your...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
Show how to synthesize the following amines from the indicated starting materials by acylation-reduction. (a) N-butylpiperidine from piperidine (b) N-benzylaniline from aniline
-
Addition of one equivalent of ammonia to 1-bromoheptane gives a mixture of heptan-1-amine, some dialkylamine, some trialkylamine, and even some tetraalkylammonium bromide. (a) Give a mechanism to...
-
Show how Gabriel syntheses might be used to prepare the following amines. (a) Benzylamine (b) hexan-1-amine (c) g-aminobutyric acid
-
Required: Following Simplicity Financial and Business Advisory Group Pty Ltd policies and procedures prepare the following: Task 1 Financial statement analysis The client needs a review of financial...
-
SCC Company reported the following for the current year.Net sales$ 57,000Cost of goods sold47, 200Beginning balance in inventory2,900Ending balance in inventory8,960Compute (a) inventory turnover and...
-
You are an assistant financial consultant for Simplicity Financial and Business Advisory Group PtyLtd, and you have been notified the client converted their business to a public company PTCG Ltd....

Study smarter with the SolutionInn App


