Propose a plausible mechanism for the following process, called iodolactonization: I2
Question:
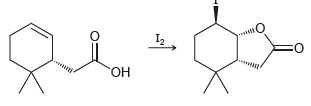
Transcribed Image Text:
I2 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)

Answered By

Muhammad Azeem Anwar
After I graduated with my bachelor's degree, I immediately started tutoring. I was interested in sharing my knowledge with others to improve their academic performance. My experience as atutor has been good, despite overcoming some few obstacles along my path. I have worked with various tutoring companies and my experience has been tremendous. I am willing to further my experience in this company. I will be dedicated to my tasks and ensure that I move the company to greater heights if given an opportunity. I will maintain close ties with the students and follow up to ensure that they succeed in their future endeavors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a plausible mechanism for the following transformation: |,+
-
Propose a plausible mechanism for the following tautomerization process: . CH CH3 ,
-
Propose a plausible mechanism for the following transformation: Br2 Br ,*
-
In an organization, managers communicate information downward to their departments and teams, and employees communicate information upward to their managers. If all members of an organization are not...
-
What are the most likely threats to the continued operation of Dirt Bikes' systems?
-
What are some unique property features of materials bonded by metallic bonds?
-
Your company is evaluating a new project, and you've estimated that the cost of capital for the project will be 8 percent. Assume that you have also projected the following cash flows for the...
-
To estimate the real rate of interest, the economics division of Mountain Banks'a major bank holding company'has gathered the data summarized in the following table. Because there is a high...
-
Compute the rate (in %) for the loan. Round answers to the nearest tenth of a percent; use ordinary interest when time is stated in days. Principal Rate (%) Time Interest $54,000 % 60 days $873
-
Green Lawns, Inc., performs adjusting entries every month, but closes its accounts only at yearend. The following is the company's year-end adjusted trial balance dated December 31, current year. a....
-
Propose a plausible mechanism for each of the following reactions: a. b. Br Br2 . [H,SO,]
-
When 3-bromocyclopentene is treated with HBr, the observed product is a racemic mixture of trans-1,2-dibromocyclopentane. None of the corresponding cis-dibromide is observed. Propose a mechanism that...
-
Exhaust (assumed to have the properties of standard air) leaves the 4-ft-diameter chimney shown in Video V5.4 and Fig. P5.42 with a speed of \(6 \mathrm{ft} / \mathrm{s}\). Because of the wind, after...
-
Suppose that, in each period, the cost of a security either goes up by a factor of u = 2 or down by a factor d = 1/2. Assume the initial price of the security is $100 and that the interest rate r is...
-
Discuss how SAA and Kenya collaboration broader growth strategy will ensure that they become a major player in regional travel
-
Lawrence Poppin, the CEO of PremiumInternetCable, has expressed great admiration for your work and has invited you to engage in another consultation session during this consultation, Mr. Poppin...
-
A small start up consulting company hopes to have 8 times the clientele it had the previous year for the next 4 years. If the company just meets its targets each of the 4 years and it had 8 clients...
-
Refer to the information for Alisha Incorporated on the previous page. However, now assume that Alisha produced 80,000 medical stints (rather than 150,000). (Note: Round all answers to two decimal...
-
A consolidated-drained triaxial test is carried out on a sand specimen that is subjected to 100 kN/m 2 confining pressure. The vertical deviator stress was increased slowly such that there is no...
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
Myrcene, C10H16, is found in oil of bay leaves and is isomeric with ?-Ocimene (Problem 14.48). It has an ultraviolet absorption at 226 nm and can be catalytically hydrogenated to yield 2,...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
Iverson, Inc. has $1,296,000 of sales revenue, $706,000 of variable costs, and $659,000 of fixed costs in 2023. Assuming the contribution margin percentage stays the same in 2024, what will the break...
-
You are considering using an asset that you did not develop, and you correctly assume that it is protected by IPR. Before using the work, what should you do? Choose all that apply and then select...
-
Cash received from a customer for unearned subscription revenue can initially be recorded as either a(n) (revenue/expense) or a(n) (liability/expense). No matter how an unearned revenue was initially...

Study smarter with the SolutionInn App


