Propose a structure for a compound with molecular formula C 8 H 18 O that exhibits the
Question:
Propose a structure for a compound with molecular formula C8H18O that exhibits the following1H NMR and13C NMR spectra.
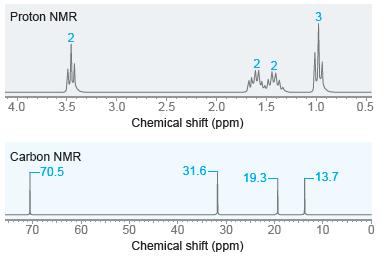
Transcribed Image Text:
Proton NMR 3.0 2.0 Chemical shift (ppm) 4.0 3.5 2.5 1.5 1.0 0.5 Carbon NMR -70.5 31.6- 19.3- -13.7 70 60 50 40 30 20 10 Chemical shift (ppm) 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)

Answered By

Seema kuldeep
although I don't have an experience of teaching in a particular institute, previously I was an expert on Chegg and I have used to teach my batch mates and also my juniors.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
Propose a structure for an alcohol with molecular formula C5H12 O that has the 1H NMR spectrum given in Fig. 9.46. Assign the chemical shifts and splitting patterns to specific aspects of the...
-
Draw a Lewis structure for a compound with molecular formula C 4 H 11 N in which three of the carbon atoms are bonded to the nitrogen atom. What is the geometry of the nitrogen atom in this compound?...
-
Mookie The Beagle Concierge Trial Balance As of January 31,2023 is given 1001 Checking 1010 Money Market 1100 Accounts Receivable (A/R) 1210 Prepaid Expenses:Supplies 1220 Prepaid Expenses:Insurance...
-
The clique problem can be stated as follows: Given an undirected graph G = (V, E) and an integer K, does G contain a complete subgraph of at least K vertices? The vertex cover problem can be stated...
-
Consider the equation y = x + 1 + 1/x-1. a. Without graphing, name the asymptotes of the function. b. Rewrite the equation as a single rational function. c. Sketch a graph of the function without...
-
Coca-Cola is considered to have one of the most valuable brand names in the world. The firm has an after-tax operating margin of 20% on revenues of \($25\) billion. The capital invested in the firm...
-
Variable and absorption costing and breakeven points Mega-Air, Inc., manufactures a specialized snowboard made for the advanced snowboarder. Mega-Air began 2011 with an inventory of 240 snowboards....
-
You invest $25,000 today at 8% per year. How much money willyou have accumulated after 13 years?You are going to receive $150,000 in 25 years. Calculate thepresent value of the $150,000 using disco 2...
-
Water is poured into a container in the shape of a right circular cone with radius 4 feet and height 16 feet. See the figure. Express the volume V of the water in the cone as a function of the height...
-
Propose a structure for ether with molecular formula C 7 H 8 O that exhibits the following 13 C NMR spectrum. Carbon NMR 114.0 129.5, 120,714.0 55.1- 159.71 160 120 140 100 100 Chemical shift (ppm)...
-
Propose a structure for a compound with molecular formula C 4 H 8 O that exhibits the following 13 C NMR and FTIR spectra. Carbon NMR 67.7- 25.4- 80 30 20 100 90 70 60 40 10 Chemical shift (ppm) 100...
-
From past experience, the owner of a restaurant knows that, on average, 4% of the parties that make reservations never show. How many reservations can the owner accept and still be at least 80% sure...
-
a. What authority underlies the Rules of the Code? b. To whom and in what circumstances do the Rules apply?
-
For what purpose does an auditor perform analytical procedures in the final stages of the audit?
-
What activities are included in departmental planning? What are the three types of planning, as discussed in Professional Standards Bulletin 83-10? Briefly discuss each of the three.
-
What is a management representation letter? Who usually prepares it? Who should sign it? When should it be dated? How does it differ from a management letter?
-
For your state, determine whether there are any laws or board of accountancy rules or regulations, other than those in a code of ethics, that deal with the matters listed below. If so, determine...
-
Find all three currents in the circuit of Fig. 25.13, but now with ? 2 = 1.0 V. E = 6 V E = 9 V R = 20R3 = 1n R2 = 40 (a) Node A, Ra (Loops Loop! Node B Loop 3 (b)
-
Define the term utility software and give two examples.
-
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain. , , . -C-C-C-
-
Construct a tree diagram for the absorption of Hm assume that Jam . . - -C-
-
Predict the multiplicities of the absorptions for the hydrogen's of these groups, assume that hydrogen's labeled a are different from those labeled x but that all of those labeled a are identical and...
-
An accountant who believes fraud is in the best interest of the company and believes everything will go back to normal after the fraud occurs is subject to Blank______. Multiple choice question....
-
Review the balance sheet for JKL Industries as of December 31, 2023, to understand its financial stability and liquidity. JKL Industries Balance Sheet As of December 31, 2023 Assets Current Assets...
-
The purpose of a pourover will is to: Multiple choice question. set up education funds for all of your surviving children. place assets into a trust at death that you may have neglected to place in...

Study smarter with the SolutionInn App


