Starting with potassium phthalimide as your source of nitrogen and using any other reagents of your choice,
Question:
Compounds in 23.18
(a)
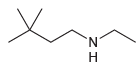
(b)
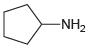
(c)
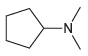
(d)
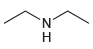
(e)
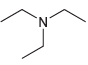
(f)
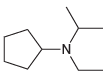
Transcribed Image Text:
-NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
The first alkyl group is installed via a Gabriel synthesis and the remaining alkyl gro...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Starting with sodium azide as your source of nitrogen and using any other reagents of your choice, show how you would prepare each of the compounds in Problem 23.18. Compounds in 23.18 (a) (b) (c)...
-
Starting with benzene and using any other reagents of your choice, show how you would prepare each of the following compounds: a. b. c. d.
-
Starting with cyclohexene and using any other reagents of your choice, show how you would prepare each of the following compounds. a. b. c. OH OMe
-
1. Given the image of the circle, identify the length of the radius, diameter, and circumference. Also, find the area. Make sure to show work (area and circumference) to receive credit. Choose a...
-
Assume that the short-run cost and demand data given in the table below confront a monopolistic competitor selling a given product and engaged in a given amount of product promotion. Compute the...
-
Draw structural representations of each of the following molecules. Give a systematic name for any compound whose name is not in accord with IUPAC nomenclature: (a) isobutylcyclopentane; (b)...
-
Oliver Scott has just made the final payment on his mortgage. He could continue to live in the home; cash expenses for repairs and maintenance (after any tax effects) would be $500 monthly....
-
Dudley Savings Bank wishes to take a position in Treasury bond futures contracts, which currently have a quote of 105-100. Dudley Savings thinks interest rates will go down over the period of...
-
Find the maximum rate of change of f(2,3,2)=2+3/2 at the point (4.-4. 1) and the direction in which it occurs. Maximum rate of changes Direction (unit vector) in which it occurs: %
-
Read the following MCQs and rectify the errors: I. The primary purpose of the statement of cash flows is to report all major cash receipts (inflows) and cash payments (outflows) during a period. True...
-
Using ammonia as your source of nitrogen, show the reagents you would use to prepare each of the following amines: (a) (b) (c) (d) (e) (f) Z
-
Suppose that the U.S. government begins charging a $1 sales tax to all consumers for each dress shirt they buy. a. What is likely to happen to the price (not including the tax) and quantity demanded...
-
The price of a bond issued by C & M plc is 85.50 per cent of par value. The bond will pay an annual 8.5 per cent coupon until maturity (the next coupon will be paid in one year). The bond matures in...
-
Why is a network control terminal a threat to the overall security of a communications network? What controls should be exercised to try to ensure that a network control terminal is used only for its...
-
What is the overall purpose of a network control terminal in a wide area communications network? Give three specific functions that can be performed by an operator using a network control terminal.
-
Why do file servers within a local area network need to be physically secured?
-
_____ allows an individual to do poorly on one test but make up for that poor grade by doing exceptionally well on other tests.
-
List two aspects of the operations of a local area network that need to be monitored to ensure that data integrity within the network is maintained. Briefly explain why these two aspects need to be...
-
For the past year, you have been working as a secretary processor for a local construction company, XYZ Homes, which specializes in the building of low-cost, limited-option homes. You left a...
-
You purchase a bond with a coupon rate of 6.7 percent, a par value $1,000, and a clean price of $905. Assume a par value of $1,000. If the next semiannual coupon payment is due in two months, what is...
-
The artificial sweetener aspartame (sidebar, p. 1208) was withheld from the market for several years because, on storage for extended periods of time in aqueous solution, it forms a diketopiperazine...
-
The artificial sweetener aspartame (sidebar, p. 1208) was withheld from the market for several years because, on storage for extended periods of time in aqueous solution, it forms a diketopiperazine...
-
The artificial sweetener aspartame (sidebar, p. 1208) was withheld from the market for several years because, on storage for extended periods of time in aqueous solution, it forms a diketopiperazine...
-
untingLab Homework: Question 1, E22-24 (similar to) Part 2 of 9 HW Score: 53.78%, 24.2 of 45 points Points: 0 of 9 Save Jefferson Labs, a taxpaying entity, estimates that it can save $29,000 a year...
-
Exercise 11-30 (Algo) Cost Allocation: Step Method (LO 11-3) Caro Manufacturing has two production departments, Machining and Assembly, and two service departments, Maintenance and Cafeteria. Direct...
-
Dahn Corporation has provided the following financial data: Balance Sheet December 31, Year 2 and Year 1 Assets Year 2 Year 1 Current assets: Cash Accounts receivable, net Inventory Prepaid expenses...

Study smarter with the SolutionInn App


