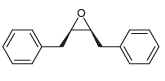
Using toluene and acetylene as your only sources of carbon atoms, show how you would prepare the
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
NBS heat MC...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
Using acetylene as your only source of carbon atoms, identify a synthetic route for the production of 2-bromobutane.
-
What are the types of production systems in aquaculture?
-
Outline the ABC inventory control procedure. What is the purpose of classifying inventory items using this procedure?
-
(a) Prove that the inverse transpose operation (1.55) respects matrix multiplication: (AB)-T = A-TB-T. (b) Verify this identity for A-C) 0
-
Calculate the following: 1. \(\left(3.6 \times 10^{-2} ight) /\left(1.5 \times 10^{3} ight)\) 2. \(\left(1.8 \times 10^{4} ight) /\left(4.8 \times 10^{3} ight)\)
-
One of New England Airs top competitive pri-orities is on- time arrivals. Quality VP Clair Bond decided to person-ally monitor New England Airs performance. Each week for the past 30 weeks, Bond...
-
I just want to get an example, of how you would do this, so that I can apply it to my paper. I know it's a minimum of 2 pages with all this information.. the goals for recruiting, recruitment...
-
An inexperienced accountant for Prestwick Company prepared the following income statement for the month of August 2015: Prepare a revised income statement in accordance with generally accepted...
-
What is a sustainable process?
-
Using the expression dS = C p /T dt VdP, calculate the decrease in temperature that occurs if 2.25 moles of water at 310. K and 1650. bar is brought to a final pressure of 1.30 bar in a reversible...
-
A study on the enhancing effect of coffee on long-term memory found that 35 participants given 200 mg of caffeine performed better on a memory test 24 hours later compared to the placebo group that...
-
Solve the following problem by using MATLAB given th 1-27 2-10 x-30 Find I Add file
-
Think about current political movements or campaigns connected to specific geographies. What kinds of economic discussions might be taking place during the upcoming primary and general elections for...
-
2. (10) In a few sentences, describe what the script below is doing (FYI: GE means greater than or equal). Be as specific as possible. UMULL RO, R1, R2, R3 CMP R1, # 0x01 BGE Elsewhere
-
An all-equity firm is considering the following projects: Project Beta IRR W .80 10.2% .90 1.10 1.35 X Y Z 11.4 12.6 15.1 The T-bill rate is 4 percent, and the expected return on the market is 12...
-
Holly Homeowner was a homeowner no more. Due to a protracted illness, she was unable to work. When her disability benefits ran out, she was unable to make her mortgage payment. Subsequently, she lost...
-
The numbers of flights by NASA astronauts, as listed in Data Set 10 in Appendix B. Refer to the indicated data set and determine whether the data have a normal distribution. Assume that this...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
Give the structure of the nucleophile that could be used to convert iodoethane into each of the following compounds in an SN2 reaction. (a) CH3CH2CN (b) CH3 alas-Cal, CH CH2S
-
. According to the salary comparison in Figure 1, by the end of a 30-year career a college graduate is likely to earn: a. $22,895. b. $40,478. c. $686,850. bd. $1,214,340.
-
Miracle Works Inc. is located in Newfoundland. The company hires 3 eligible apprentices, paying each of them $30,000 in the current taxation year. It also acquires $2,000,000 in qualified depreciable...
-
Shareholders' equity and liabilities both have normal credit balances. Why are the shareholders' equity debit/credit rules more complex than liabilities? O The elements of Shareholders' Equity are...

Study smarter with the SolutionInn App


