Valine has the structure Draw the zwitterion that would exist at neutral pH. H CH3CH-C-COOH - CH3
Question:
Valine has the structure
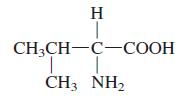
Draw the zwitterion that would exist at neutral pH.
Transcribed Image Text:
H CH3CH-C-COOH -СООН CH3 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Zwitterion ...View the full answer

Answered By

Himanshu Kumar
I have worked for various online sites as a doubt solver and subject experts .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Alanine has the structure Draw the zwitterion that would exist at neutral pH. CH3-C-COOH NH
-
Which one of the following five structures is a zwitterion? (a) (b) - O 2 CCH 2 CH 2 CO 2 - (c) H 3 N + CH 2 CO 2 - (d) CH 3 (CH 2 ) 16 CO 2 - K + (e) 0,CCH,CNH,
-
Consider the fully protonated amino acid valine: where the numbers denote the pKa values. (b) Calculate the predominant form of valine at pH 1.0, 7.0, and 12.0. (c) Calculate the isoelectric point of...
-
The top 5 stocks in the S&P 500 index, when ranked by market capitalization, make up 22% of the total market capitalization of the S&P 500 index. Numerical estimates of the mean (or expected) rates...
-
The number of automobile accidents reported per month helps to identify intersections that require improvement. The number of crashes per month reported at an intersection near a university campus in...
-
On October 1, Dalton Corp. borrows \(\$ 100,000\) from National Bank, signing a six-month note payable for that amount, plus interest to be computed at a rate of 9 percent per annum. Indicate all...
-
It is \(3: 00\). What time of the day will it be in 150 hours?
-
A comparative balance sheet for Orozco Corporation is presented below. Additional information:1. Net income for 2012 was $105,000.2. Cash dividends of $40,000 were declared and paid.3. Bonds payable...
-
Carnes Cosmetics Co.'s stock price is $42, and it recently paid a $2.75 dividend. This dividend is expected to grow by 22% for the next 3 years, then grow forever at a constant rate, g; and r s =...
-
A 2-in.-OD stainless steel tube has 16 longitudinal fins spaced around its outside surface as shown. The fins are 1/16-in. thick and extend 1 in. from the outside surface of the tube. a. If the...
-
Consider the following polymer: From which two monomers is the polymer made? -C(CH2)10NH(CH2)6 NHC(CH2)10CNH(CH2)6NH-
-
If a sample of DNA isolated from a microorganism culture were analyzed and found to contain 1.5 mol of cytosine nucleotides and 0.5 mol of adenosine nucleotides, what would be the amounts of guanine...
-
Discuss the key aspects of designing product portfolios and product lines.
-
1. What is meant by "improved decision-making"? Explain the ways in which operations management can contribute to this. Provide examples if deemed necessary. 2. What are the differences between terms...
-
Problem 2: Euler's Method Approximation (25 points) For the function f(x) given in Problem 1, we split the interval [1,4] into the sub-intervals [1, 2], [2, 3], and [3,4]. On each sub-interval, we...
-
what is expected from a regulatory standpoint when looking for approval of combinamt product classified as class 3 device using nanotechnology
-
As the stakeholders provide their needs and expectations to improve the process, identify steps that could be improved with the support of a hiring system. Also look for examples of issues and...
-
When a dermatologist suspects that skin may show signs of disease or cancer they will likely take a skin biopsy for analysis. This process involves removing a small portion of the affected skin. In a...
-
Gus, a football player who was renegotiating his contract with the Denver Broncos, paid his ex-girlfriend $50,000 to drop a sexual assault complaint against him and keep the matter confidential. The...
-
What is a make-or-buy decision?
-
A piston cylinder contains 3 kg of air at 20oC and 300 kPa. It is now heated up in a constant pressure process to 600 K.
-
An insulated cylinder is divided into two parts of 1 m3 each by an initially locked piston. Side A has air at 200 kPa, 300 K, and side B has air at 1.0 MPa, 1000 K. The piston is now unlocked so it...
-
A piston cylinder contains air at 600 kPa, 290 K and a volume of 0.01 m3. A constant pressure process gives 54 kJ of work out. Find the final temperature of the air and the heat transfer input.
-
A capital loss would be allowed on the sale of: Multiple Choice a). equipment included in Class 8. b). shares which were then reacquired 15 days later. c). shares of a small business corporation...
-
A disgusted or hopeless consumer is more likely to complain than an angry consumer. a . True b . False
-
ENGR 3040 ASSIGNMENT: Case Study 2 LOCATION DUE: Canvas Dropbox EFFORT: Individual POINTS: 100 GRADING CATEGORY: Case Studies DESCRIPTION: In March 2019, aviation authorities worldwide grounded the...

Study smarter with the SolutionInn App


