When the following compound is treated with Br 2 in the presence of a Lewis acid, one
Question:
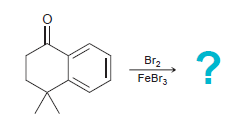
Transcribed Image Text:
Br FeBr3 ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
Br...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What product is formed when the following compound is treated with Ag2O? HO
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
Compare the hoop stress (r,/2) predictions from elasticity with voids given by relation (15.5.18) with the corresponding results from classical theory. Choosing N = 1/2 and L = 2, for the elastic...
-
How can market research benefit a small business owner? List some possible sources of market information.
-
A new dormitory is being built at a college in North Carolina. To save costs, it is proposed to not include air conditioning ducts and vents. A member of the board overseeing the construction says...
-
On March 1, 20XA, Chuck Larson purchases mineral rights (MR) for $30,000. On June 1, 20XA, he leases the mineral rights to Grey Wolf Oil Company, retaining a 1/5 royalty interest (RI). Grey Wolf Oil...
-
Prime Company holds 80 percent of Lane Company's stock, acquired on January 1, 20X2, for $160,000. On the date of acquisition, Lane reported retained earnings of $50,000 and $100,000 of common stock...
-
On January 1, 2018, you deposited $6,600 in a savings account. The account will earn 12 percent annual compound interest, which will be added to the fund balance at the end of each year. Required: 1....
-
The condensed financial statements of Murawski Company for the years 2021 and 2022 are presented as follows. (Amounts in thousands.) Compute the following ratios for 2022 and 2021. a. Current ratio....
-
Ca(HCO 3 ) 2 (s) decomposes at elevated temperatures according to the stoichiometric equation Ca(HCO 3 ) 2 (s) ???? CaCO 3 (s) + H 2 O(g) + CO 2 (g). a. If pure Ca(HCO 3 ) 2 (s) is put into a sealed...
-
Calculate A for the isothermal compression of 2.95 mol of an ideal gas at 325 K from an initial volume of 60.0 L to a final volume of 20.5 L. Does it matter whether the path is reversible or...
-
Exhibit contains the income statements and balance sheets of The Hershey Company for the years ended December 31, 2011, and December 31, 2010. Hershey manufactures and sells products under more than...
-
Others always ask me why do you believe in him 'I don't know that's all that's really why, why do I agree? Sweet words Like latik not cooked in milk You know but with a different face Why, why do I...
-
At December 31, 2023, Cord Company's plant asset and accumulated depreciation and amortization accounts had balances as follows: Category Land Land improvements Buildings Equipment Automobiles and...
-
Good Scent, Inc., produces two colognes: Rose and Violet. Of the two, Rose is more popular. Data concerning the two products follow: Rose Violet Expected sales (in cases) 51,000 10,200 Selling price...
-
San Antonio Premier Car Services provides a variety of maintenance services for vehicles. Ricardo placed an order in March 2024 for a new set of tires for $395. When a customer purchases goods or...
-
Cash Budgeting Collections during month of sale Collections during 1st month after sale Collections during 2nd month after sale General and administrative salaries Lease payments 10% 75% 15% $33,000...
-
Visit http://www.qualitymag.com, search for "statistical quality control," find an application, and summarize it.
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
What is the wavelength of light with a wave-number of 1720 cm-1?
-
Predict the relative intensities of the three peaks in the mass spectrum of dichloromethane at m/z = 84, 86, and 88.
-
A single-step income statement shows only one subtotal for expenses.True false question.TrueFalse
-
Agito and its subsidiaries (Agito Group) are principally engaged in the production and distribution of high-density polyethylene (HDPE) in Hong Kong. On 1 April 2022, Agito signed an agreement with...
-
Problem 10-14 (Algo) Measures of Internal Business Process Performance [LO10-3] DataSpan, Incorporated, automated its plant at the start of the current year and installed a flexible manufacturing...

Study smarter with the SolutionInn App


