Estimate the heat of vaporization (kJ/mol) of benzene at 25C, using each of the following correlations and
Question:
Estimate the heat of vaporization (kJ/mol) of benzene at 25°C, using each of the following correlations and data:
(a) The heat of vaporization at the normal boiling point and Watson’s correlation.
(b) The Clausius–Clapeyron equation and boiling points at 50 mm Hg and 150 mm Hg.
(c) Tables B.1 and B.2.
(d) Find a tabulated value of the heat of vaporization of benzene at 25°C. (Suggestion: Do the same thing you do when you want to find almost any item of information.) Then calculate the percentage errors that result from the estimations of Parts (a), (b), and (c).
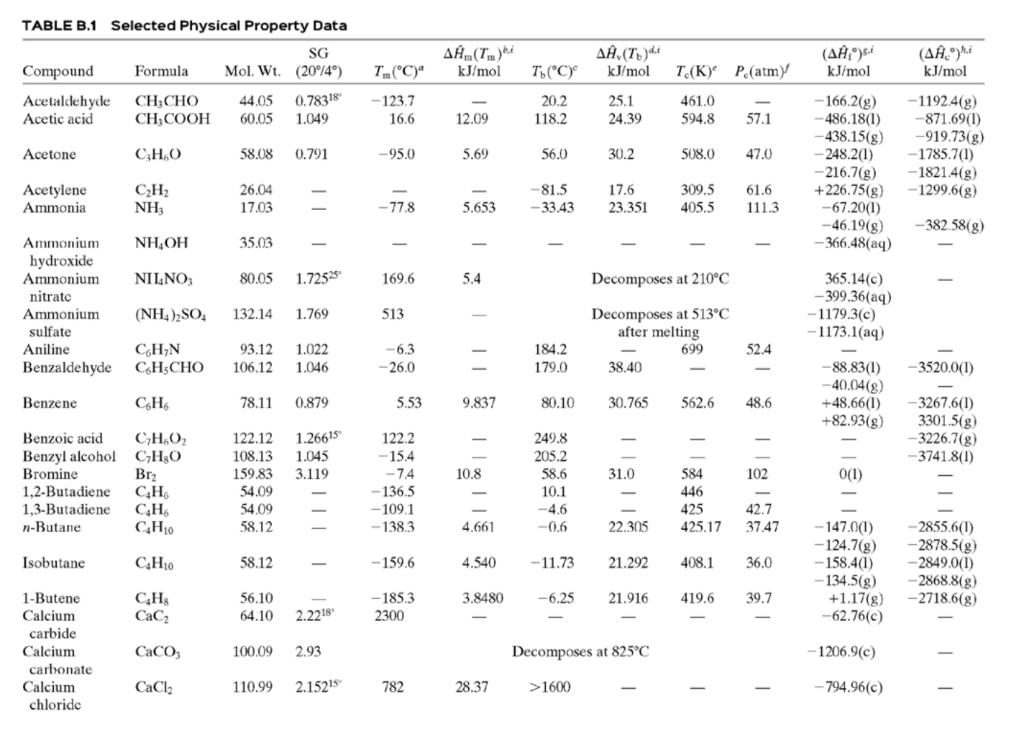
Table B.2
![TABLE B.2 Heat Capacities Form 1: C,[kJ/(mol-C)] or [kJ/(mol-K)] = a + bT + cr + dT Form 2: C,k/(molC)j or [kJ/(mol-K)j = a + bT + cT2 Example: (C,)acetone(e) = 0.07196 + (20.10 x 10-5)r - (12.78 x 10-8)7? + (34.76 x 10-12)Tr*, where T is in °C. %3D](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1590/0/8/2/6645ec6bc68ba9271590082649906.jpg)
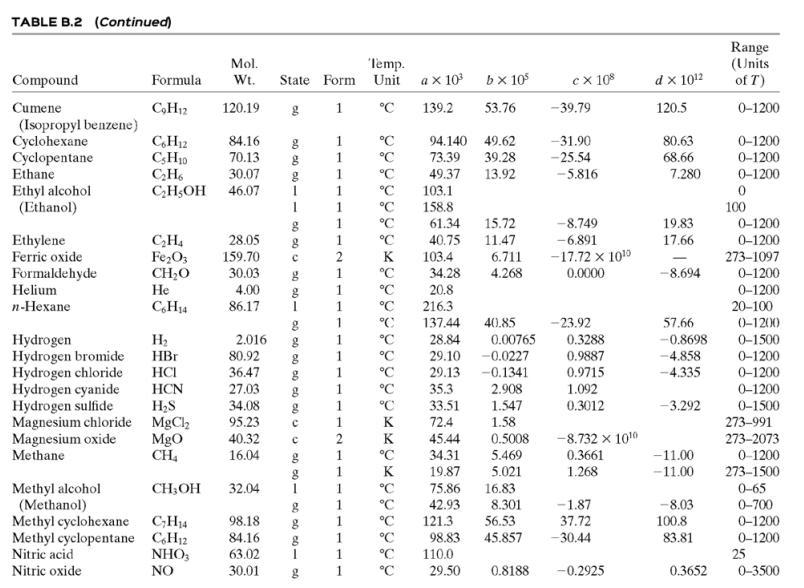
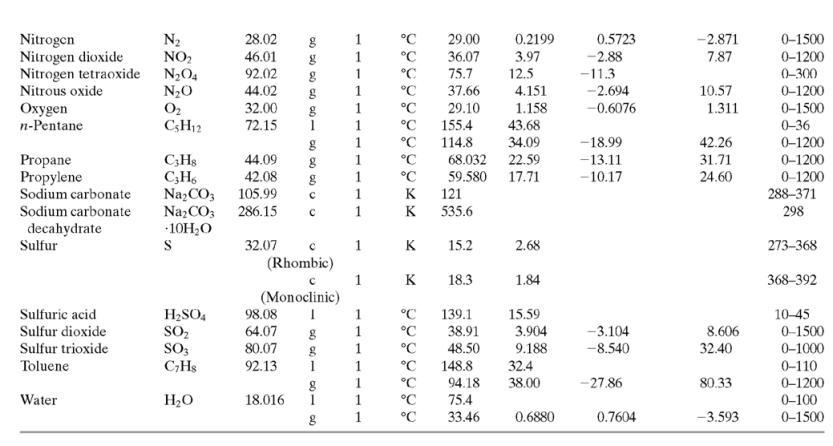
TABLE B.2 Heat Capacities Form 1: C,[kJ/(mol-C)] or [kJ/(mol-K)] = a + bT + cr + dT Form 2: C,k/(molC)j or [kJ/(mol-K)j = a + bT + cT2 Example: (C,)acetone(e) = 0.07196 + (20.10 x 10-5)r - (12.78 x 10-8)7? + (34.76 x 10-12)Tr*, where T is in °C. %3D Note: The formulas for gases are strictly applicable at pressures low enough for the ideal-gas equation of state to apply. Range (Units of T) Mol. Temp. State Form Unit a x 10 ex 10 d x 1012 Compound Formula Wt. bx 10 Acetone CH;COCH; 58.08 1 °C 123.0 18.6 -30-60 °C 71.96 34.76 20.10 6.053 0.4147 -12.78 -5.033 0.3191 0-1200 0-1200 0-1500 C;H2 26.04 29.0 °C 42.43 28.94 28.09 18.20 -1.965 1.965 -6.686 Acetylene Air 0.1965 0.4799 273-1800 Ammonia NH, 17.03 1 °C 35.15 2.954 0.4421 0-1200 Ammonium sulfate Benzene (NH4),SO4 132.15 C,H, 215.9 275-328 6-67 126.5 74.06 78.11 1 °C 23.4 32.95 -25.20 77.57 0-1200 CH10 CH10 CHs CaC2 CACO, Ca(OH)2 CaO Isobutane 58.12 89.46 49.87 30.13 27.88 -18.91 0-1200 0-1200 n-Butane 58.12 92.30 -15.47 34.98 Isobutene 56.10 82.88 25.64 -17.27 50.50 0-1200 Calcium carbide Calcium carbonate Calcium hydroxide 64.10 68.62 -8.66 x 1010 -12.87 x 1010 2 K 1.19 298-720 100.09 K 82.34 89.5 4.975 273-1033 74.10 K 276-373 -4.52 x 1010 -4.891 x 1010 -2.887 Calcium oxide 56.08 12.01 K 41.84 2.03 273-1173 Carbon с 11.18 1.095 273-1373 CO2 CO Carbon dioxide 44.01 36.11 4.233 7.464 0-1500 Carbon monoxide 28.01 1. 28.95 0.4110 0.3548 -2.220 0-1500 Carbon tetrachloride CC4 Ch 153.84 93.39 12.98 1.367 0.6117 273-343 0-1200 273-1357 Chlorine 70.91 1 °C 33.60 -1.607 6.473 Соpper Cu 63.54 1. K 22.76 212 21-
Step by Step Answer:
a b c d The heat of vaporization at normalt b p of benzene is 3072 kJm2 ieat 1009c 35...View the full answer



Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Related Video
The experiment aims to show the impact of various beverages on teeth by using eggs as a representation of enamel. Three eggs are boiled and then placed in glasses filled with fizzy drinks, vinegar, and mango juice for 24 hours. The shells of eggs are similar to enamel as they are composed of calcium carbonate, and enamel is primarily made of calcium phosphate. The eggs are then observed to demonstrate the effects of the different liquids on teeth and the importance of brushing regularly. The egg placed in fizzy drink has turned dark in color but can be cleaned by brushing with toothpaste and rinsing with water. The egg placed in vinegar has had its shell softened due to the chemical reaction of vinegar and calcium carbonate, which can\'t be reversed. This highlights the fact that acids are more damaging to teeth than other substances. The egg placed in mango juice represents the process of bacteria in the mouth converting sugars and starches into acids that form plaque, which can be prevented by brushing. The use of fluoride in toothpaste is also highlighted as it slows down the demineralization process and protects the enamel. The importance of brushing teeth twice a day is emphasized.
Students also viewed these Sciences questions
-
Estimate the heat of vaporization (kJ/mol) of benzene at a pressure of 100 mm Hg, using each of the following correlations and data: (a) The heat of vaporization at the normal boiling point given in...
-
Estimate the heat of vaporization of ethyl benzene at its normal boiling point using Troutons rule and Chens rule and compare the results with a tabulated value of this quantity. Then estimate Hv at...
-
The heat of vaporization of water at the normal boiling point, 373.2 K, is 40.66 kJ/mol. The specific heat capacity of liquid water is 4.184 JK-1g-1 and of gaseous water is 2.02 J K-1g-1. Assume that...
-
Using Table 1.6, write a structural formula for each of the following: a. An alcohol, C3H8O b. An ether, C4H10O c. An aldehyde, C3H6O d. A ketone, C3H6O e. A carboxylic acid, C3H6O2 f. An ester,...
-
Given the following reactions and their enthalpies: a. Devise a way to calculate H for the reaction H2O(g) 2H(g) + O(g) b. From this, estimate the H - O bond energy. (kJ/mol) +436 +495 H2(g)- 2H(g)...
-
Square Root (10 points) Given a natural number n, create an algorithm based on binary search to compute the square root of n (the biggest natural number k such that k2 2.) Give pseudo-code for your...
-
Few board members were born with natural fundraising skills. What are the most effective ways to involve every board member in fundraising?
-
1. Columbiana is a small, independent island in the Caribbean that has many historical buildings, forts, and other sites, along with rain forests and striking mountains. A few first- class hotels and...
-
35 0.56 points Please read these survey items and answer the following questions. Item Item Text Strongly Agree Neutral Disagree Strongly Agree Disagree A People convicted of murder or rape should be...
-
Karen Most has a federal tax levy of $2,100.50 against her. If Most is single with two personal exemptions and had a take-home pay of $499.00 this week, how much would her employer take from her pay...
-
In gas adsorption a vapor is transferred from a gas mixture to the surface of a solid. (See Section 6.7.) An approximate but useful way of analyzing adsorption is to treat it simply as condensation...
-
A gas stream containing n-hexane in nitrogen with a relative saturation of 90% is fed to a condenser at 75C and 3.0 atm absolute. The product gas emerges at 0C and 3.0 atm at a rate of 746.7 m 3 /h....
-
Why are information systems vulnerable to destruction, error, and abuse? 1. List and describe the most common threats against contemporary information systems. 2. Define malware and distinguish among...
-
A distillation column is operating under a vacuum. The column has 18 stages with the feed at stage 9, a partial reboiler, and a partial condenser. The pressure drop is 5.0 mm Hg in the condenser, 4.0...
-
For a simple, single-feed distillation column, derive an equation for calculation of \((\overline{\mathrm{V}} / \mathrm{B})_{\min }\) from \((\overline{\mathrm{L}} / \overline{\mathrm{V}})_{\max }\)
-
What should the internal auditor include in an individual, tailor-made program?
-
In a well-managed EDP installation, the internal auditor should expect to find: a. A password protection system. b. Duplicate files for all computer systems. c. Computer operators with an extensive...
-
How is net sales computed?
-
The following transactions of Austin's Marine Supply occurred during 2015 and 2016: 2015 Feb 3 Purchased equipment for $15,000, signing a six-month, 4% note payable. 28 Recorded the week's sales of...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Most common amines (RNH 2 ) exhibit pK a values between 35 and 45. R represents the rest of the compound (generally carbon and hydrogen atoms). However, when R is a cyano group, the pK a is found to...
-
Provide a systematic name for each of the following compounds: a. b. c. d.
-
Compound X has molecular formula C 7 H 14 . Hydrogenation of compound X produces 2, 4-dimethylpentane. Hydroboration-oxidation of compound X produces a racemic mixture of 2, 4-dimethylpentan-1-ol...
-
Keiko is buying a used car. Payments will be $ 7 8 . 7 5 every week for 2 years, with the first payment at the end of 1 3 weeks. The interest rate is 7 . 0 7 5 % compounded monthly. What is the...
-
Sosa Company has $39 per unit in variable costs and $1,900,000 per year in fixed costs. Demand is estimated to be 138,000 units annually. What is the price if a markup of 35% on total cost is used to...
-
Do you think employers have a right to check into applicants' backgrounds? What about if that check includes social media, blogs, pictures, or writings that can be found on the internet? Mountain Ski...

Study smarter with the SolutionInn App


