The following data are for the chemisorption of hydrogen on copper powder at 25 C. Confirm that
Question:
The following data are for the chemisorption of hydrogen on copper powder at 25 °C. Confirm that they fit the Langmuir isotherm at low coverages. Then find the value of α for the adsorption equilibrium and the adsorption volume corresponding to complete coverage.
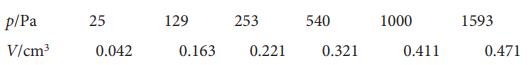
Transcribed Image Text:
p/Pa V/cm³ 25 0.042 129 0.163 253 0.221 540 0.321 1000 0.411 1593 0.471
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Calculations The Langmuir isotherm is given by PP01PP0 where fractional coverage P pressure ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The following data are for Ernst Company. Note: All inventory is purchased on account, and Accounts Payable relates only to the purchase of inventory. Instructions: Compute the following: 1. The...
-
The following data are for a simple steam power plant as shown in Fig. P6.39 State 1 2 3 4 5 6 7 P MPa 6.2 6.1 5.9 5.7 5.5 0.01 0.009 T C 45 175 500 490 40
-
The following data are for a simple steam power plant as shown in Fig. P6.39 State 2 3 4 5 P psia 890 830 1.4 900 860 800 1.5 TF 115 350 920 900 110 6.
-
Telstar Limited was incorporated on 2 January 2015. On 3 January 2015, the company ordered plant from Germany at a cost of E1 million. The plant was loaded free on board in Hamburg on 1 March 2015,...
-
Data set: Tennis Students on the college tennis team were interested in knowing if string tension affected the speed and accuracy of a tennis ball. They had recently read an article in the Journal of...
-
John Dear Co. is a heavy equipment and lawn care products manufacturer located in Illinois. John Dear is publicly owned, and has declared the following dividends over a six year period: 20X1, $...
-
4. YOU BE THE JUDGE WRITING PROBLEM Asher Hyman and Stephen Stahl formed a corporation named Ampersand to produce plays. Both men were employed by the corporation. After producing one play, Stahl...
-
Journal entries, T-accounts and source documents. Production Company produces gadgets for the coveted small appliance market. The following data reflects activity for the year 2008. Production Co....
-
Mark has recently begun working at a local fruit and vegetable grocer. Mark spends a good deal of his time talking to customers and fulfilling sales orders. Over time, he identifies a weakness in ...
-
A number of corporations today are working to reengineer corporate thinking and encourage an innovative environment. What types of steps would you recommend? Offer at least three and explain each.
-
Drawing from knowledge you have acquired through the text, describe the advantages and limitations of each of the microscopy, diffraction, and ionizations techniques designated by the acronyms AFM,...
-
The volume of oxygen gas at 0 C and 104 kPa adsorbed on the surface of 1.00 g of a sample of silica at 0 C was 0.286 cm 3 at 145.4Torr and 1.443 cm 3 at 760Torr. What is the value of V mon ?
-
Compute y and dy for the given values of x and dx = x. Then sketch a diagram like Figure 5 showing the linesegments with lengths dx, dy, and y. y = x 2 - 4x, x = 3, x = 0.5
-
A statistical analysis of 1 , 0 0 0 long - distance telephone calls made by a company indicates that the length of these calls is normally distributed, with mean = 2 2 0 seconds and deviation = 4 0...
-
Andretti Company has a single product called a Dak. The company normally produces and sells 88,000 Daks each year at a selling price of $58 per unit. The company's unit costs at this level of...
-
What role does Facebook and Twitter play in consumer word-of-mouth? What is buzz marketing and how is buzz marketing related to consumer word-of-mouth? What buzz marketing campaigns are you familiar...
-
Electron Inc. produces a variety of electronic products which it sells to retail stores throughout the country. The following data is available for the year for one of the products: Units started...
-
textbook lists five components necessary for effective auditing and monitoring of risk management and governance processes. Explain the components and identify a relevant or applicable AICPA Code for...
-
An asset used in a four-year project falls in the five-year MACRS class for tax purposes. The asset has an acquisition cost of $6,500,000 and will be sold for $1,600,000 at the end of the project. If...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
Describe the model used to determine the heat capacity of atomic crystals.
-
What is the Boltzmann formula, and how can it be used to predict residual entropy?
-
How does the Boltzmann formula provide an understanding of the third law of thermodynamics?
-
Review | Constants An oil tanker spills a large amount of oil (n = 1.45) into the sea (n = 1.35). (a) If you look down onto the oil spill from overhead, what predominant wavelength of light do you...
-
Lees Home Goods, which sells home decor items, had $150,000 of cost of goods sold during the month of October. The company projects an 8 percent increase in cost of goods sold during November. The...
-
Review | Constants Monochromatic light of wavelength 580 nm passes through a single slit and the diffraction pattern is observed on a screen. Both the source and screen are far enough from the slit...

Study smarter with the SolutionInn App


