The vapor pressure of a liquid can be written in the empirical form known as the Antoine
Question:
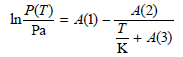
Starting with this equation, derive an equation giving ΔH Vaporization m as a function of temperature.
Transcribed Image Text:
A(2) A(1) – - + A(3) P(T) In- т Pa к
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
For a liquidgas equilibrium ...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The conversion of the kinetic energy of wind to electricity may be an attractive alternative to the use of fossil fuels. Typically, wind causes the rotor of a turbine to turn, and a generator...
-
The Arnold Diffusion Cell shown in Figure 26.5 is a simple device used to measure gas-phase diffusion coefficients for volatile substrates in air. In the present experiment, liquid acetone is loaded...
-
In this problem, you calculate the error in assuming that ÎH o R is independent of T for the reaction 2CuO(s) 2Cu(s) + O 2 (g). The following data are given at 25°C:
-
Problem 9- 3A Estimating and reporting bad debts P2 At December 31, 2013, Hawke Company reports the following results for its calendar year. Cash sales . . . . . . . . . . $ 1,905,000 Credit sales ....
-
1. Using ERG theory, explain the reasons for the situation described in the case. Existence needs (E): Relatedness needs (R): Growth needs (G): 2. Using expectancy theory, explain the reasons for the...
-
An air compressor has an isentropic efficiency of 0.88. If air at 100 kPa and 25C and a mass ow rate of 20 000 kg/hr is compressed to 500 kPa, what work must be supplied to the compressor? Assume...
-
The Canada Business Corporations Act requires all com panies incorporated under it to have audit committees. Required a. Describe an audit committee. b. What are the typical functions performed by an...
-
Holz Disc Golf Course was opened on March 1 by Ian Holz. The following selected events and transactions occurred during March. Mar. 1 Invested $20,000 cash in the business. 3 Purchased Rainbow Golf...
-
10 6 points Silva Chairs, Incorporated makes two types of chairs. Model Diamond is a high-end product designed for professional offices. Model Gold is an economical product designed for family use....
-
Izabela Jach opened a medical office under the name Izabela Jach, MD, on August 1, 2017. On August 31, the balance sheet showed Cash $3,000; Accounts Receivable $1,500; Supplies $600; Equipment...
-
Calculate the vapor pressure for a mist of spherical water droplets of radius a. 1.95 10 8 m b. 2.25 10 6 m at 298 K. The vapor pressure of water at this temperature is 25.2 Torr.
-
Use the following vapor pressures of propane given here to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P (Torr) T (K) 0.01114 100. 120 2.317...
-
A long, straight wire carrying a current of 5 A flowing from left to right is placed above a large, flat sheet through which the current per unit width is \(6.0 \mathrm{~A} / \mathrm{m}\) flows from...
-
Use the income statement and the list of changes to answer the question. Dansko Integrated Income Statement January 1 to December 31, 2022 (amounts in thousands) Revenue 8,500 Cost of Goods Sold...
-
The electric field midway between two equal but opposite point charges is 286 N/C, and the distance between the charges is 15.0 cm. Part A What is the magnitude of the charge on each? Express your...
-
a. Bob invests $11,500 in a 4 year Certificate of Deposit (CD) at the bank with an interest rate of 5.5%. Using simple interest with an annual period, what will the CD be worth at the maturity date?...
-
Valerie: As you know, we considered 3 cities and are leaning towards Norman. What do you think? Sara :The Workforce Planning data shows a considerable difference in the number of competitors in each...
-
Aram's taxable income before considering capital gains and losses is $83,000. Determine Aram's taxable income and how much of the income will be taxed at ordinary rates in each of the following...
-
The current assets and liabilities sections of the comparative balance sheets of Regent Inc., a private entity reporting under ASPE, at December 31 are presented below, along with the income...
-
Match the following. Answers may be used more than once: Measurement Method A. Amortized cost B. Equity method C. Acquisition method and consolidation D. Fair value method Reporting Method 1. Less...
-
Find theLaplace transform of the function f(t) = t n e at , where n is an integer.
-
Find the Laplace transform of sin 2 (at).
-
Derive the version of Eq. (11.49) for n = 2.
-
D . 1 , Il and III. Lily is a college student who likes to buy two goods: Cheetos and Pepsi. Her utility for these two goods depends on I. Lily's preferences for Cheetos and Pepsi. II . The prices of...
-
Part B: Category vs. function (intermediate) [11 points] The -ly adverbs (adjective type modificational items in non-adjective positions) are just one type of item that has an adverbial function. The...
-
igure 7 - 1 1 nar 0 0 2 - 1 . jpg Refer to Figure 7 - 1 1 . If the government imposes a price ceiling of $ 7 0 in this market, then the new producer surplus will be a . $ 3 5 0 . b . $ 1 7 5 . c . $...

Study smarter with the SolutionInn App


