Fractional composition in a diprotic system. Create a spreadsheet with Equations 9-19 through 9-21 to compute the
Question:
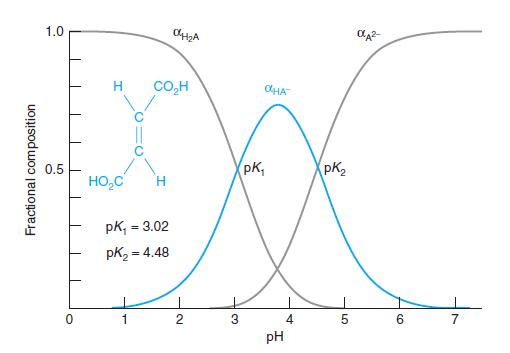
Fractional composition in a diprotic system. Create a spreadsheet with Equations 9-19 through 9-21 to compute the three curves in Figure 9-4. Plot the three curves in a beautifully labeled figure.
Figure 9-4
Equations 9-19 to 9-21

Transcribed Image Text:
1.0 H CO,H OHA pK, PK, 0.5 HO,C pk, = 3.02 %3! pK2 = 4.48 2 3 4 5. 6. 7 pH Fractional composition
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
pH alphaH2A alphaHA alphaA2 0 0999046 0000954 316E08 0070707 0998877 0001123 437E08 0141414 0998679 0001321 606E08 0212121 0998446 0001554 839E08 0282828 0998172 0001828 116E07 0353535 0997849 0002151 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Fractional composition in a triprotic system. For a triprotic system, the fractional composition equations are where D = [H + ] 3 + K 1 [H + ] 2 + K1K 2 [H + ] + K 1 K 2 K 3 . Use these equations to...
-
Fractional composition in a tetraprotic system. Prepare a fractional composition diagram analogous to Figure 9-4 for the tetraprotic system derived from hydrolysis of Cr3+: Cr3+ + (H2O) Cr(OH)2+ +...
-
A solution containing 0.139 mmol of the triprotic acid tris(2-aminoethyl)amine 3HCl plus 0.115 mmol HCl in 40 mL of 0.10 M KCl was titrated with 0.490 5 M NaOH to measure acid dissociation...
-
Alpha Mann Ltd makes and sells computer carry bags. Bill Blake, the company accountant, is responsible for preparing the company's annual budget. In compiling the budget data for next year, Blake has...
-
Dr. Tooth is a 40-year-old dentist in Small Town, USA. He graduated from dental school five years ago and has had a thriving practice ever since. At the end of last year, however, Dr. Tooth had an...
-
The following information is taken from the records of Cramer Corporation on March 31 of the current year. The accounts and balances needed to complete this problem are provided in the Working...
-
What qualitative factors should you consider regarding accepting an order to sell Sears the boats for less than the price you charge your regular customers? For example, what would your regular...
-
Bavarian Chocolate Company processes chocolate into candy bars. The process begins by placing direct materials (raw chocolate, milk, and sugar) into the Blending Department. All materials are placed...
-
Points: 0 of 1 Save You would like to buy a house that costs $350,000. You have $50,000 in cash that you can put down on the house, but you need to borrow the rest of the purchase price. The bank is...
-
Your Competitive Intelligence team reports that a wave of product liability lawsuits is likely to cause Digby to pull the product Dart entirely off the market this year. Assume Digby scraps all...
-
Calculate H2A , HA -, A2- and for cis-butenedioic acid at pH 1.00, 1.92, 6.00, 6.27, and 10.00.
-
Explain what is wrong with the following statement: At its isoelectric point, the charge on all molecules of a particular protein is 0.
-
The amount of milk (in thousands of gallons) that a dairy sells each week is a random variable X with the density function f (x) = 4(x - 1)3, 1 x 2. (See Fig. 4.) (a) What is the likelihood that...
-
Nike, the global athletic footwear market is expected to experience only very slow growth over the next several years. Nike is the market leader. According to Boston Consulting Group portfolio...
-
Solve the following using related rates and show all steps of working 1. A cylindrical tank with radius5m is being filled with water at a rate of 2m 3 /min. How fast is the height of the water...
-
A rectangular poster is to contain 512 square inches of print. The margins at the top and bottom of the poster are to be 2 inches, and the margins on the left and right are to be 1 inch. What should...
-
What are four key policies, programs and/or initiatives that led to healthcare reform in the US healthcare system. You will need to specifically identify (by name) and describe each of the four key...
-
Abe has led multiple teams that are too cohesive. He has decided he needs someone who will be willing to be a devils advocate on his next team. What follower type is likely to take on and be the...
-
Zevo Chemical Company Zevo is currently reaching the end of the requirements modeling phase of a new inventory control system for their global company. As a systems analyst, you are asked to support...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
Cable AB on the boom truck is hoisting the 2500-lb section of precast concrete (Figure P4.18). A second cable is under tension P, and workers use it to pull and adjust the position of the concrete...
-
Solve the problem of Example 4.5 by using the force components method. Replace the polar representation of the anchor straps tension by the horizontal and vertical components T x and T y , and solve...
-
A front loader of mass 4.5 Mg is shown in side view as it lifts a 0.75-Mg load of gravel (Figure P4.20). (a) Draw a free body diagram of the front loader. (b) Determine the contact forces between the...
-
Listen The desire of foreign producers to preserve market share for goods sold in the United States helps contribute to complete exchange-rate pass-through following a depreciation of the dollar True...
-
You work at a large department store selling computer products. Iwina walks in and wants to buy a wireless router. She explains that the media streaming device she ordered online supports a...
-
Discuss the security issues surrounding the ability of users to access the network from a remote location. Explain the remote access technologies, protocols, and options. Compare VPN technology from...

Study smarter with the SolutionInn App


