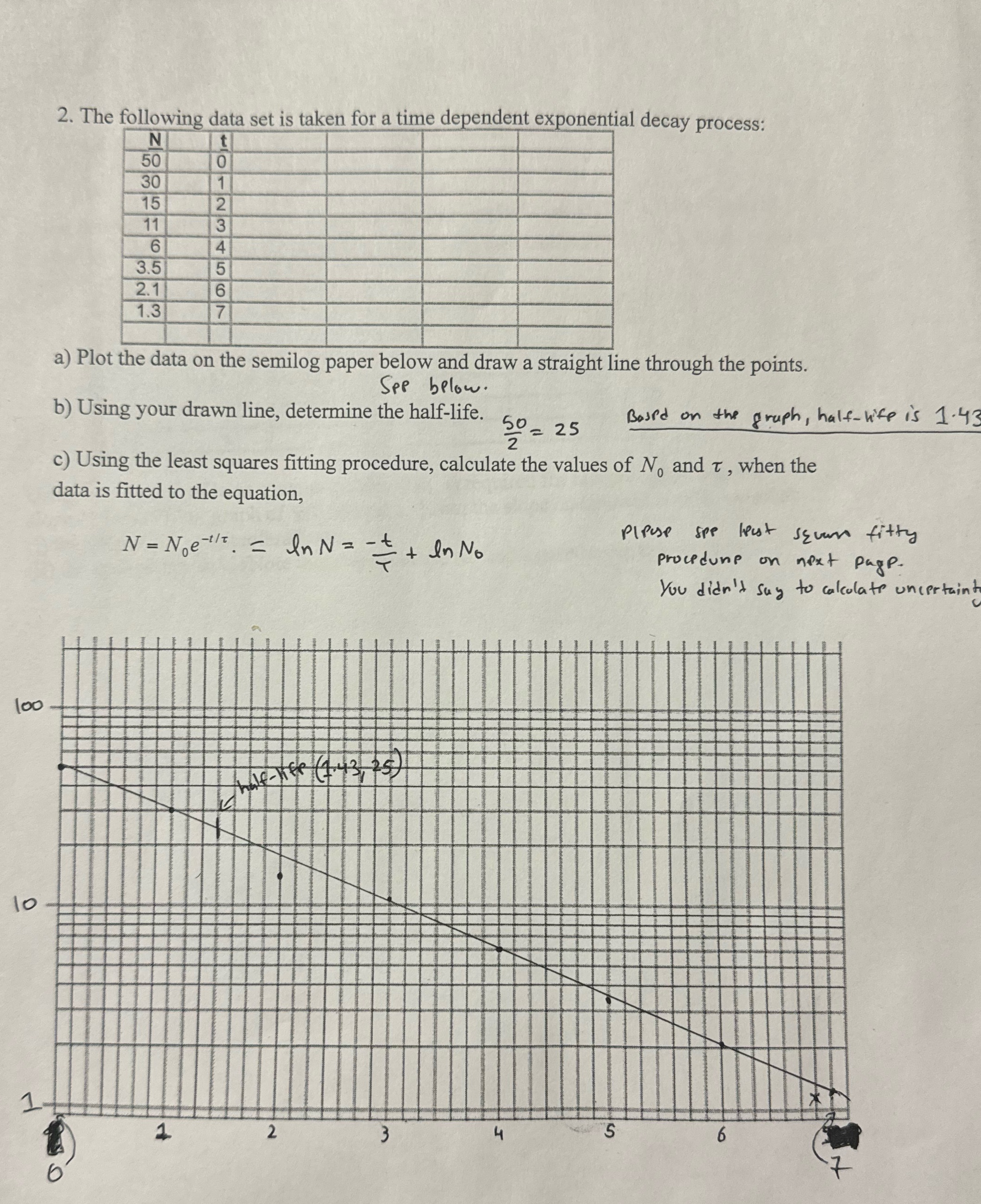
10 1 100 2. The following data set is taken for a time dependent exponential decay...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
10 1 100 2. The following data set is taken for a time dependent exponential decay process: N 30 606165 t 11 3.5 2.1 1.3 01234567 a) Plot the data on the semilog paper below and draw a straight line through the points. See below. 20=25 b) Using your drawn line, determine the half-life. Based on the graph, half-life is 1.43 c) Using the least squares fitting procedure, calculate the values of N, and T, when the data is fitted to the equation, = N = Noe In N = In N = = + + In No 1 Pleuse Spe least seven fitty next pagp. Procedune on next You didn't say to calculate uncertaint half-life (1.43, 25) 1 2 3 4 S 9 10 1 100 2. The following data set is taken for a time dependent exponential decay process: N 30 606165 t 11 3.5 2.1 1.3 01234567 a) Plot the data on the semilog paper below and draw a straight line through the points. See below. 20=25 b) Using your drawn line, determine the half-life. Based on the graph, half-life is 1.43 c) Using the least squares fitting procedure, calculate the values of N, and T, when the data is fitted to the equation, = N = Noe In N = In N = = + + In No 1 Pleuse Spe least seven fitty next pagp. Procedune on next You didn't say to calculate uncertaint half-life (1.43, 25) 1 2 3 4 S 9
Expert Answer:

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these physics questions
-
Archeologists have discovered a rise in shaft graves during the Middle Helladic period in ancient Greece (i.e., around 2000 BC). Shaft graves are named for the beautifully decorated sword shafts that...
-
You may need to use the appropriate appendix table or technology to answer this question. A sample of 81 patients in a doctor's office showed that they had to wait an average of 49 minutes with a...
-
CANMNMM January of this year. (a) Each item will be held in a record. Describe all the data structures that must refer to these records to implement the required functionality. Describe all the...
-
For the manipulator shown, find the twist coordinates f; at zero con- figuration and 9st (0) using the product of exponentials approach. Use the base and tool frames shown. (b) (10 points) Find efi0i...
-
Majestic America Transportation (MAT) began 20X6 with accounts receivable, inventory, and prepaid expenses totaling $65,000. At the end of the year MAT had a total of $78,000 for these current...
-
Sara's trial balance at 31 March 2004 was as follows. Required Prepare Sara's Trading and Profit and Loss Account for the year ended 31 March 2004. Account $ loc binoin Cash on kont Sales Stock...
-
Question: Douglas Kuroda worked for the Hertz Corp. He and his supervisor had a heated argument in which Kuroda told his boss, "You may have a master's degree but you don't know shit." The supervisor...
-
On December 21, 2012, Zurich Company provided you with the following information regarding its trading investments. During 2013, Carolina Company shares were sold for $9,500. The fair value of the...
-
At the beginning of current year, Pedro Company reported fair value of plan assets at P6,500,000 and projected benefit obligation at P7,500,000. During the current year, the entity determined that ...
-
Translate the following call to Schemes COND to C and set the resulting value to y. (COND ( (> x 10) x) (( < x 5) (* 2 x)) ( (= x 7) (+ x 10))
-
python coding language 1. Binomial distribution (20 + 10 points) Write a program tosimulate an experiment of tossing a fair coin 16 times and countingthe number of heads. Repeat this experiment 10**5...
-
The solubility of magnesium hydroxide, Mg(OH)2, in water is 9.0 x10^-4 g/100 mL. What volume of 0.00600 M HNO3 is required to neutralize 1.00 L of saturated Mg(OH)2 solution?
-
Potassium chloride (KCI) has a molar mass of 74.551 g/mol. Calculate the mass in grams of KCl required to prepare 50.00 mL of a 0.300 mol/L aqueous solution of KCl. Report your answer to the correct...
-
Your very smart colleague Andres Parra Munoz has been making elaborate nots for Test 2. One part of his notes reads as follows: 'Demnding domestic buyers for industry's products ssuper greater...
-
Ten grams of a sample of metal is added to 50.0 mL of water in agraduated cylinder. The final volume in the graduate is 52.8 mL.Calculate the density of the metal.
-
Visible light falls into wavelength ranges of 400-700 nm, forwhich 1 m = 1 10 9 nm . The energy and wavelength of light arerelated by the equation E = h c ? where E is energy in Joules, h...
-
While formal contracts are carefully drafted to ensure that each of these elements are fully incorporated in the contract docs, they may have no more validity than contracts drafted on the "back of...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
During 2012, Tom sold Sears stock for $10,000. The stock was purchased 4 years ago for $13,000. Tom also sold Ford Motor Company bonds for $35,000. The bonds were purchased 2 months ago for $30,000....
-
Karim Depak received a Form 1099-B showing the following stock transactions and basis during 2012: None of the stock is qualified small business stock. Calculate Karim's net capital gain or loss...
-
Charlie's Green Lawn Care is a cash basis taxpayer. Charlie Adame, the sole proprietor, is considering delaying some of his December 2012 customer billings for lawn care into the next year. In...
-
B Mann has the following purchases for the month of May 19X8: Required: (a) Enter up the purchases journal for the month. (b) Post the transactions to the suppliers accounts. (c) Transfer the total...
-
A Rowland has the following purchases for the month of June 19X9: Required: (a) Enter up the purchases journal for the month. (b) Post the items to the suppliers accounts. (c) Transfer the total to...
-
C Phillips, a sole trader, has the following purchases and sales for March 19X9: Required: (a) Prepare the purchases and sales journals of C Phillips from the above. (b) Post the items to the...

Study smarter with the SolutionInn App


