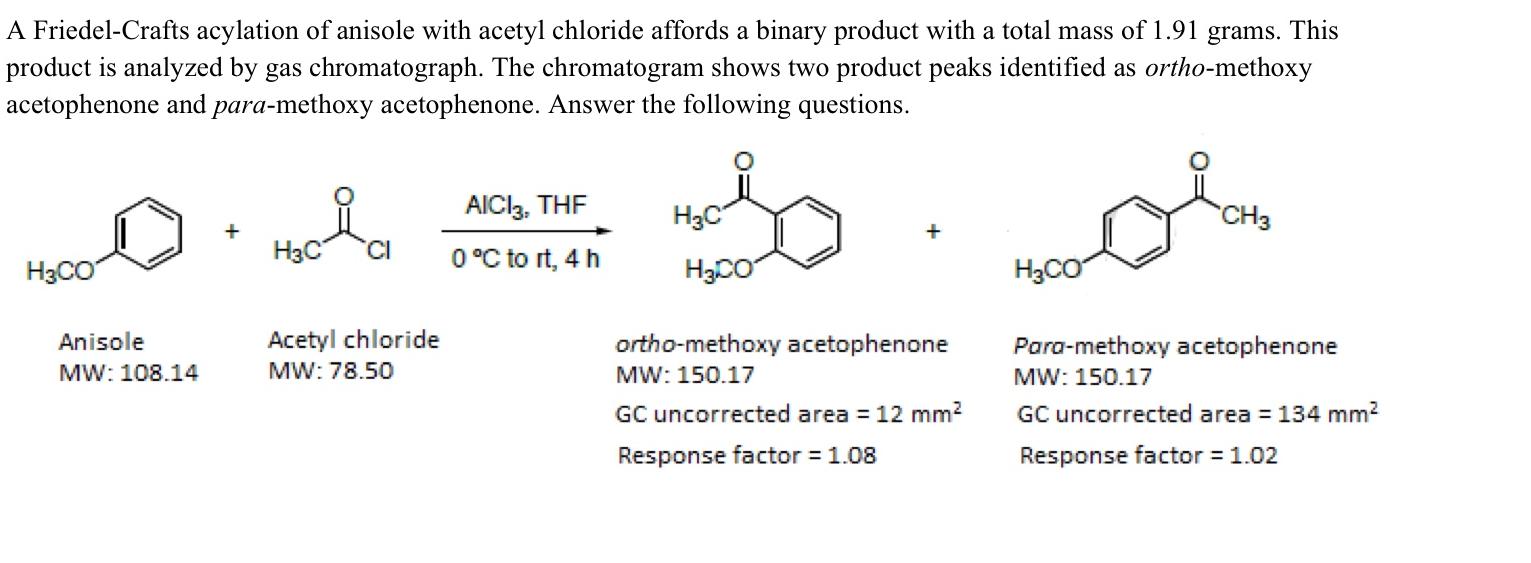
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas. A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas.
Expert Answer:
Answer rating: 100% (QA)
2 A Given total mass or the product area 12 mm Para pordnet area a 134 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
The results taken from a mass spectrum of chlorine gas show peaks at m/z 35.00 and m/z 37.00 (The m/z peaks on a mass spectrum identify the different isotopes of an element that are present in the...
-
A rectangular aluminum bar 12 mm thick and 50 mm wide is welded to fixed supports at the ends, and the bar supports a load W = 3.5 kN, acting through a pin as shown. Find the reactions at the...
-
1-Calculate amount of secondary compression in terms of inches 10 years following end of the primary consolidation. S.sc ang (12)= Coefficient of secondary compression=0.035 Thickness of clay layer-...
-
What is an angel investor? Assemble a brief profile of the typical private investor. How can entrepreneurs locate potential angels to invest in their businesses?
-
During the year, Summit produces 40,000 snow shovels and sells 37,000 snow shovels. Required What is variable cost of goods sold?
-
Use the information from question 7. When the merchandise is sold, at what amount will the company report cost of goods sold? Use the following forward and spot prices for Canadian dollars (C\($)\)...
-
Danielle Manning, D.D.S., opened a dental practice on January 1, 2012. During the first month of operations, the following transactions occurred. 1. Performed services for patients who had dental...
-
AAA is a medical company and must decide between two mutually exclusive projects for the final year of its life. The two projects have different levels of risk and their ultimate payoff depends on...
-
A village has six residents, each of whom has accumulated savings of $100. Each villager can use this money either to buy a government bond that pays 15 percent interest per year or to buy a year-old...
-
. Question 1 a} Explain what is meant by a perfect hedge. Does a perfect hedge always lead to a better outcome than an imperfect hedge? (10 marks} b} An investor pays $2 for a call with strike pri...
-
What is the condition on efficiency for a machine to be reversible ?
-
What is sudden load?
-
In a differential pulley block, the diameter of the smaller pulley is 9/10 th of the larger pulley. Find the load lifted by the block with an effort of 200 N if the efficiency is 45%.
-
Show that the competitive labor market compensates workers for the probability that they will be laid off.
-
Define creep. What are its stages?
-
Assume that actual overhead is $834,000 in a given year, the overhead rate is $17 per unit, 53,000 units were sold, and 52,000 units were produced. For the end of the year, is overhead underapplied...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
As a method for the preparation of alkenes, a weakness in the acid-catalyzed dehydration of alcohols is that the initially formed alkene (or mixture of alkenes) sometimes isomerizes under the...
-
Select the combination of alkyl bromide and potassium alkoxide that would be the most effective in the syntheses of the following ethers: (a) CH3OC(CH3)3 (b) (c) (CH3)3CCH2OCH2CH3 OCH
-
Write chemical equations for the reaction that takes place between each of the following pairs of reactants: (a) 3-Ethyl-3-pentanol and hydrogen chloride (b) 1-Tetradecanol and hydrogen bromide
-
Motive to commit fraud usually will include all of the following except: a. Feelings of resentment b. Alcohol, drug, or gambling addiction c. Financial pressures and personal habits d. Inadequate...
-
Which statement is false with respect to backdating of stock options? a. Steve Jobs, CEO of Apple, was involved with backdating. b. Manipulating a stock option exercise date to a lower price will...
-
Which would not be an indicator of alter ego? a. Financial dependence behaviors b. Confusion about corporate identity c. Risk assessment d. Dominance and control e. None of the above

Study smarter with the SolutionInn App


