1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia...
Fantastic news! We've Found the answer you've been seeking!
Question:

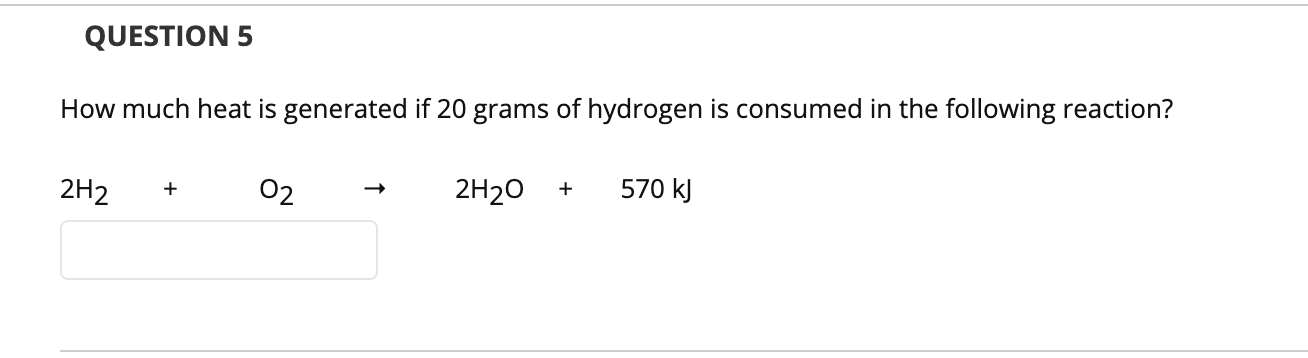
Transcribed Image Text:
1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ 1. Balance the following equations C7H14 02 CO2 H20 PCI3 H20 H3PO3 HCI QUESTION 2 Ammonia reacts with diatomic oxygen to form nitrogen monoxide and water vapor: NH3 + 02 ® NO H20 When 40.0 g NH3 and 50.0 g 02 are allowed to react, what is the theoretical yield of this reaction? If 30 g of NO is produced, what is the % yield? QUESTION 3 Aqueous solutions of sodium sulfide and lead (II) nitrate are combined. The products are sodium nitrate and lead (II) sulfide. Write the net ionic equation. Na2s + Pb(NO3)2 → 2 NaNO3 PbS s2. (aд) + Pb2+, (aq) + 2NO3 (aq) 2NO3'aq) 2 Na' (aq) PbS(s) + + + (aq) 2 Na"(aq) + 2NO3 aq) - 2 NANO3(s) s2- (aq) Pb2+, PbS(s) + (aq) QUESTION 4 What element is oxidized in the chemical reaction Cu + HNO3 → Cu(NO3)2 + NO + H20 Cu The nitrogen of the HNO3 is oxidized The hydrogen of the HNO3 is oxidized The oxygen of the HNO3 is oxidized QUESTION 5 How much heat is generated if 20 grams of hydrogen is consumed in the following reaction? 2H2 02 2H20 570 kJ
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Write the net ionic equation for CaCl2(aq) + Pb(NO3)2(aq) ( Ca(NO3)2(aq) + PbCl2(s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
Blaine is a practicing dentist. He operates his business from the basement of his house, with a separate entrance and facilities. Blaine uses the simplified method every year to calculate his...
-
Give an example of and discuss a stock that has temporary, supernormal growth where it would be appropriate (necessary) to use the modified DDM.
-
In this exercise, we will examine hill climbing in the context of robot navigation, using the environment in Figure as an example. a. Repeat Exercise 3.16 using hill climbing. Does your agent ever...
-
What can be done to stimulate creativity in decision making?
-
Based on the manager identified in Question 3, in what ways did that person exhibit or fail to exhibit each of the components of leadermember exchange (LMX)? Did you have a high or low LMX with that...
-
Entry for cash sales; cash over The actual cash received from cash sales was $8,667 and the amount indicated by the cash register total was $8,651. Journalize the entry to record the cash receipts...
-
Kaitlin Carlton, a CPA sole practitioner, prepares tax returns each year for approximately 100 clients. Items 1 through 8 each represent an independent factual situation in which Kaitlin has prepared...
-
Find tan 0, where 0 is the angle deci
-
Can you elucidate the molecular mechanisms underlying the phenomenon of epigenetic inheritance and its implications in the regulation of gene expression across successive generations ?
-
You enter into a long position in one gold futures contract worth $500 per ounce. Contract size is 100 ounces. The initial margin is $2,500 per contract and the maintenance margin is $1,500 per...
-
The concept of shareholdervaluemaximization focuses on maximizing the value of equity.But in a finance balance sheet, we can see that thisis not quite the same as maximizing the value offirm? Why?...
-
Hired an administrative assistant at an annual salary of $33,000. The administrative assistant will start working on November 1 and will be paid on the last day of the month, starting in November....
-
A candidate or member is least likely violating the Standard regarding the con!dentiality of client information if he shares con!dential client information, when not required by law, with: A) the CFA...
-
Dacosta Corporation had only one job in process on May 1. The job had been charged with $1,850 of direct materials, $6,990 of direct labour, and$9,950 of manufacturing overhead cost. The company...
-
Propose a reasonable mechanism for the following reaction. OH
-
What is the freezing point of an aqueous solution that boils at 105.0 oC?
-
Specify what ions are present in solution upon dissolving each of the following substances in water: (a) ZnCl2 (b) HNO3 (c) (NH4)2SO4 (d) Ca(OH)2.
-
Detailed calculations show that the value of Zeff for the outermost electrons in Si and Cl atoms is 4.29+ and 6.12+, respectively. (a) What value do you estimate for Zeff experienced by the outermost...
-
What factors should be taken into account when designing a system of accounting for inflation?
-
Historical cost accounting is the worst possible accounting convention, until one considers the alternatives. Discuss this statement in relation to CPP, CCA and NRVA.
-
Discuss the effect on setting performance bonuses for staff if financial performance for a period contains both realised and unrealised gains/losses.

Study smarter with the SolutionInn App



