The cyclization reaction shown below involves two sequential condensation reactions, and can be catalyzed by either...
Fantastic news! We've Found the answer you've been seeking!
Question:
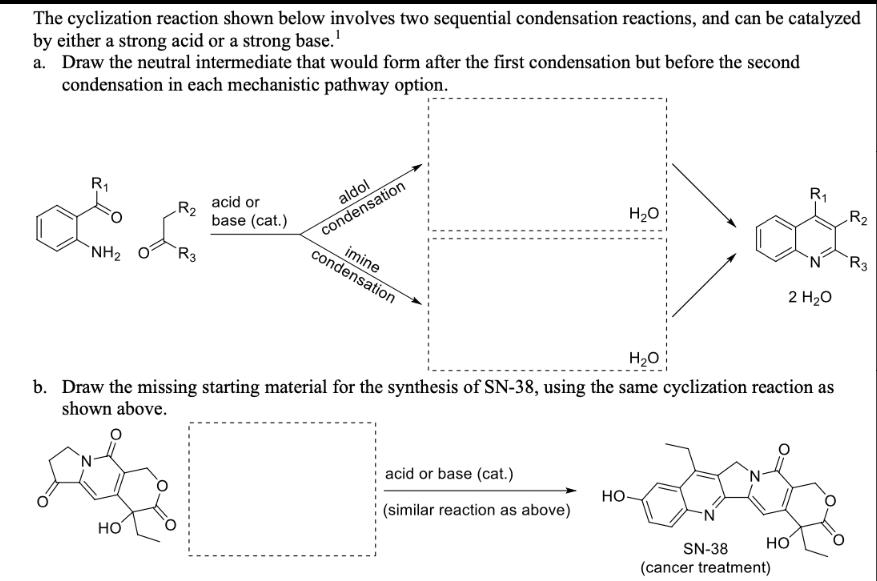
Transcribed Image Text:
The cyclization reaction shown below involves two sequential condensation reactions, and can be catalyzed by either a strong acid or a strong base.¹ a. Draw the neutral intermediate that would form after the first condensation but before the second condensation in each mechanistic pathway option. acid or R2 base (cat.) aldol condensation NH2 R3 condensation imine R₁ R2 H₂O N R3 2 H₂O H₂O b. Draw the missing starting material for the synthesis of SN-38, using the same cyclization reaction as shown above. HO acid or base (cat.) HO (similar reaction as above) HO SN-38 (cancer treatment) The cyclization reaction shown below involves two sequential condensation reactions, and can be catalyzed by either a strong acid or a strong base.¹ a. Draw the neutral intermediate that would form after the first condensation but before the second condensation in each mechanistic pathway option. acid or R2 base (cat.) aldol condensation NH2 R3 condensation imine R₁ R2 H₂O N R3 2 H₂O H₂O b. Draw the missing starting material for the synthesis of SN-38, using the same cyclization reaction as shown above. HO acid or base (cat.) HO (similar reaction as above) HO SN-38 (cancer treatment)
Expert Answer:
Answer rating: 100% (QA)
Solutions Step 1 a The starting materials for the basecatalyzed cyclization reaction of cabotegravir are 124Dichlorophenyl4ethyl13dihydro2Hbenzimidazol2one Ethyl cyanoacetate Sodium hydride NaH Dimeth... View the full answer

Related Book For 

Precalculus Concepts Through Functions A Unit Circle Approach To Trigonometry
ISBN: 9780137945139
5th Edition
Authors: Michael Sullivan
Posted Date:
Students also viewed these chemical engineering questions
-
Which two experiments are best used to determine the order of reaction for Cl2? By best, we mean "easiest." Experiment Initial [CO] (M) Initial [Cl2] (M) Initial Rate (M/s) 1 0.12 0.20 0.121 2 0.24...
-
We sell 5 0 . 0 0 0 units of a special material using 5 % of the capacity. There is a new order consisting 3 0 . 0 0 0 units from a foreign market for 6 0 0 $ . The managers of the business suggested...
-
The value of Kc for the following reaction at 900oC is 0.28. What is Kp at this temperature? CS2(g) +4H2(g)CH4(g) + 2H2S(g)
-
1. About 99 percent of the world's population lives outside Canada, but many Canadian companies, especially small businesses, still do not engage in global trade. Why not? Do you think more small...
-
Ethically, should gays and lesbians be protected against discrimination under Tide VII?
-
15 in 3. Una barra uniforme de 45Kg y 2mts de longitud gira con una velocidad angular de w = 3 rad/seg en sentido horario. La barra est sujeta a un resorte en su extremo con una constante k = 140 N/m...
-
Describe a conflict that may arise between the shareholders of a company and the debtholders of the company. Discuss what the debtholders might do to resolve that conflict and explain why you feel...
-
a. If a firm buys under terms of 3/15, net 45, but actually pays on the 20th day and still takes the discount, what is the nominal cost of its non-free trade credit? b. Does it receive more or less...
-
In the last 2 years, we were having COVID-19 pandemic. The air freight cost has increased 5-10 times, the company is questioning why you still use air freight mode. As the logistic manager of a...
-
A process is in control with x = 100.s = 1.05, and n = 5. The process specifications are at 95 ± 10. The quality characteristic has a normal distribution. (a) Estimate the potential...
-
GameWorld Co. Game World Co. was one of the largest chains of video game rental stores in the greater Boston area, operating 17 wholly owned outlets. Begun in 1998 as a small store in Coolidge Corner...
-
Derive the stopping rule for investments in education.
-
Some people claim that by increasing depreciation, firms may save corporate taxes. Do you agree? Why, or why not?
-
Give conditions for OWC to approximately be receivables plus inventory less payables.
-
Discuss how the wageschooling locus is determined in the labor market, and why it is upward sloping and concave.
-
Discuss why there is regression toward the mean in the correlation between the earnings of parents and children.
-
Predict the transition metal-containing products of the following reactions: a. cis-Mn(CO) (13CO) (COCH) (show all expected products, percentage of each) hv b. C6H5CH2-Mn(CO) 5 CO+ c. V(CO)6 + NO...
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
Problems 77 86. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for later sections, a final exam, or subsequent courses such as calculus....
-
Problems 95 104. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for later sections, a final exam, or subsequent courses such as calculus....
-
In Problems 2734, graph each function by adding y-coordinates. f (x) = x cos x
-
In an increasing-cost industry, an unexpected increase in demand would lead to what result in the long run? a. Higher costs and a higher price b. Higher costs and a lower price c. No change in costs...
-
What is allocative efficiency?
-
Describe and explain the concept of diminishing marginal product.

Study smarter with the SolutionInn App


